MDA-MB-231 xenograft model
MDA-MB-231 cells have several properties that make them useful for research. Firstly, they are relatively easy to culture in the laboratory and can be grown in large quantities. Secondly, they express markers of breast cancer, such as estrogen receptor (ER), progesterone receptor (PR), and HER2, and exhibit several features characteristic of breast cancer, including uncontrolled proliferation, invasion, and metastasis. Thirdly, they respond to various stimuli, such as cytokines and growth factors, making them a useful model system for studying the effects of these factors on breast cancer cell growth and survival.
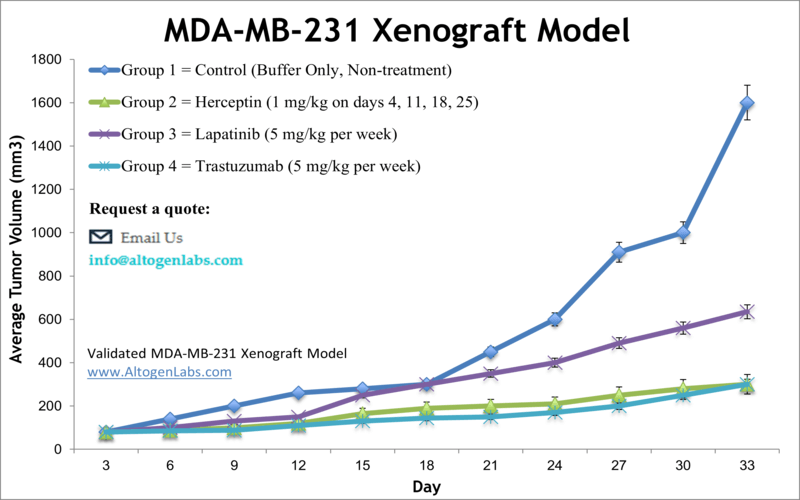
Breast cancer is the most prevalent form of cancer among American women, and has the second highest mortality rate in females after lung cancer. The MDA-MB-231 epithelial triple negative breast cancer (TNBC) cell line is isolated from a 51-year-old Caucasian female with a metastatic mammary adenocarcinoma and is very widely-used in cancer research laboratories. The MDA-MB-231 cell line is ER-negative with an invasive phenotype an appearance of spindle-shaped cells. According to the study published in Breast Disease Journal, the TNBC cell lines are extremely aggressive and mirror the original tumors, making them powerful tools for investigative breast cancer research using MDA-MB-231 xenograft model. The MDA-MB-231 cell line is used to create the CDX (Cell Line Derived Xenograft) MDA-MB-231 xenograft mouse model. The MDA-MB-231 xenograft model is HER2 negative, CK18 and EGFR positive and exhibits tumor growth inhibition from herceptin, lapatinib and trastuzumab.
Research that has used these cells include the study by Iorns et al. (2012) that examined the MDA-MB-231 xenograft mouse model to determine the expression changes that occur when breast cancer cells metastasize to from the mammary pad primary tumor to distant organs. They found that the tumor cells had several changes in gene expression that suggests its necessity for successful metastasis and secondary organ tumor growth. A 2008 study by Radestock et al. selected the MDA-MB-231 xenograft model to study the effects of relaxin on tumor cells in an oestrogen receptor alpha-negative system. Relaxin overexpression was previously associated with promotion of migration however this study identified a novel mechanism of action where long-term relaxin expression leads inhibition of growth and invasion of breast cancer cells through the down-regulation of S100A4 (metastasin), a promotor of metastasis linked to poor prognosis in cancer patients. Finally, a 2016 Nature study (Wang et al.) used the MDA-MB-231 xenograft model to investigate the mechanism of action of SL4, a compound known to slow tumor invasion and angiogenesis. They report that SL4 inhibits proliferation through cell cycle arrest with clinically relevant IC50 values and apparent toxicity.
Download Altogen Labs MDA-MB-231 Xenograft Model PowerPoint Presentation: ![]()
Basic study design
- Cells are continually grown under aseptic conditions prior to collection and in an exponential phase.
- Post- collection, cells viability is determined. A 98% minimum viability via trypan blue exclusion assay is required.
- All cell suspension concentrations are adjusted by volume such that a 100 µL injection of the Matrigel + MDA-MB-231 cell suspension contains one million cells. The cell suspension is inoculated s.c. into a flank of one hind leg per. NOD/SCID or athymic BALB/C are used and are 10-12 weeks old.
- Calipering of tumors determines start of the study, with an expectation of 50-150 mm3 Sorting of animals into cohorts and administration of test compounds are performed according to the established treatment schedule.
- Daily tumor measurements are recorded. The tumor size limit in the study plan determines the completion of the study. Necropsies are performed for tumor removal, weights are recorded and then documented digitally.
- Tumors and tissues can be snap frozen in Liquid Nitrogen, stabilized in RNAlater or nucleic acids isolated.
Get Instant Quote for
MDA-MB-231 Xenograft Model
Xenograft animal models are used to assess the effectiveness of drugs against specific types of cancer. New medicines are tested on staged tumor growths that have been engrafted via subcutaneous or orthotopic inoculation in an immunocompromised mouse or rat model. All clinically approved anti-cancer agents have been evaluated with conventional preclinical in vivo models. Xenograft studies can be highly complex, starting with the selection of the appropriate animal model, choice of tumorigenic cell line, administration method, dosing, analysis of tumor growth rates and tumor analysis (histology, mRNA and protein expression levels).
Altogen Labs provides an array of laboratory services using over 90 standard Cell Line Derived Xenograft (CDX) models and over 30 PDX models. Researchers investigating the role of specific proteins or gene products in regulating tumor growth can benefit from development of protein overexpression (genetically engineered to ectopically express proteins, tumor suppressors, or oncogenes) and RNAi cell lines with long term gene silencing. Altogen Labs provides quantitative gene expression analysis of mRNA expression (RT-PCR) and protein expression analysis using the WES system (ProteinSimple).
Animal handling and maintenance at the Altogen Labs facility is IACUC-regulated and GLP-compliant. Following acclimation to the vivarium environment, mice are sorted according to body mass. The animals are examined daily for tumor appearance and clinical signs. We provide detailed experimental procedures, health reports and data (all-inclusive report is provided to the client that includes methods, results, discussion and raw data along with statistical analysis). Additional services available include collection of tissue, histology, isolation of total protein or RNA and analysis of gene expression. Our animal facilities have the flexibility to use specialized food or water systems for inducible gene expression systems.
Following options are available for the MDA-MB-231 xenograft model:
- MDA-MB-231 Tumor Growth Delay (TGD; latency)
- MDA-MB-231 Tumor Growth Inhibition (TGI)
- Dosing frequency and duration of dose administration
- Dosing route (intravenous, intratracheal, continuous infusion, intraperitoneal, intratumoral, oral gavage, topical, intramuscular, subcutaneous, intranasal, using cutting-edge micro-injection techniques and pump-controlled IV injection)
- MDA-MB-231 tumor immunohistochemistry
- Alternative cell engraftment sites (orthotopic transplantation, tail vein injection and left ventricular injection for metastasis studies, injection into the mammary fat pad, intraperitoneal injection)
- Blood chemistry analysis
- Toxicity and survival (optional: performing a broad health observation program)
- Gross necropsies and histopathology
- Positive control group employing cyclophosphamide, at a dosage of 20-40 mg/kg administered by intramuscular injection to the control group daily for the study duration
- Lipid distribution and metabolic assays
- Imaging studies: Fluorescence-based whole body imaging
