Preclinical Studies: Xenograft (efficacy) and Pharm/Tox (safety)
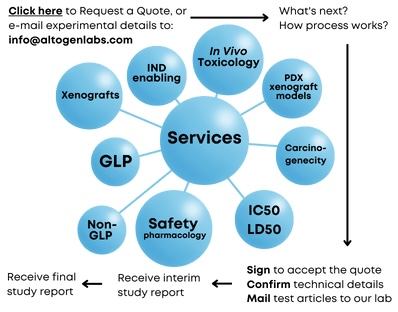
Altogen Laboratories, located in Austin, TX, is a GLP-compliant laboratory that provides preclinical research services for pharmaceutical, biotechnology, and academic institutions worldwide. We provide biology CRO in vivo services, including 98 in-house validated CDX models, 47 PDX xenograft models, and 14 syngeneic murine models. Our services include safety in vivo toxicology studies (for IND applications), efficacy xenograft studies (for regulatory submissions), drug discovery (patent applications), drug development, and other types of R&D studies including efficacy, safety toxicology, biodistribution, and imaging studies. Preclinical research studies often require a sequence of experiments utilizing various scientific methods to demonstrate test article efficacy, safety, and mechanism of action. Altogen scientists have years of experience and expertise that includes all types of nonclinical contract research studies, cell and molecular biology R&D services, pharmacology, and toxicology safety studies that are required for IND. Our in vitro services include 28-day development of stable cell lines, testing compound efficacy and toxicity, gene function and gene expression analysis by qPCR (mRNA) and automated Western Blot system Wes (protein expression), RNAi laboratory services, transient and stable transfections, test compound liposome encapsulation for tissue-targeted delivery, and more.
Technical support and QA/QC teams ensure the company’s high-quality preclinical efficacy and toxicology services. We support clients from the study design phase through patent or regulatory submissions (100% of IP belongs to the client). Altogen Labs offers instant quotes, comprehensive service packages, and competitive pricing. We provide a wide range of GLP laboratory services to meet clients’ unique needs, including stable cell line development in 28 days, A-to-Z complete RNAi services (from oligonucleotide design to tissue-targeted delivery), anticancer compound testing, liposome encapsulation (proteins, plasmid DNA, si/shRNA, mRNA, and small molecules), cryopreservation, and cell banking services. We are happy to discuss your experimental details to help generate accurate price quotes and timeline estimates (please e-mail study details to info@altogenlabs.com, or call us at 512-433-6177).