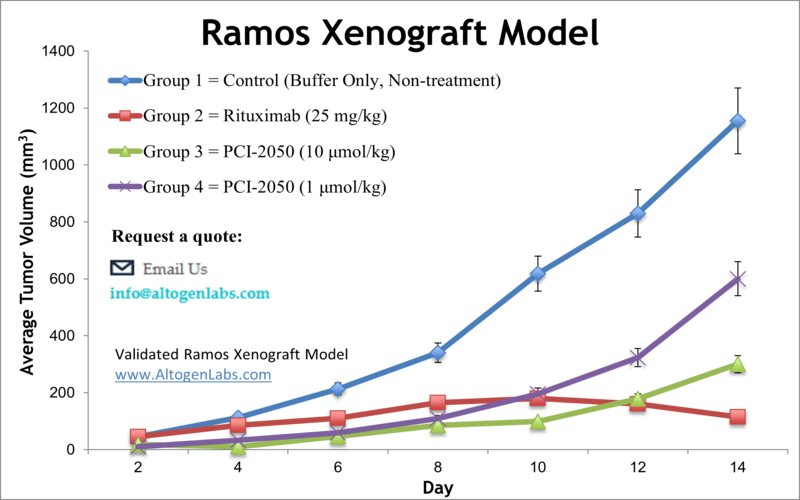
Ramos xenograft model
Ramos cells are a type of B lymphocyte cell line that is commonly used in research as a model for studying B cell biology and diseases. The Ramos cell line was derived from a Burkitt lymphoma patient and is known to express a number of important B cell markers, including CD19, CD20, CD22, and immunoglobulin M (IgM). These cells are often used to study the mechanisms of B cell activation, differentiation, and apoptosis, as well as to develop new therapies for B cell-related diseases such as lymphoma and leukemia. Burkitt’s lymphoma is a form of non-Hodgkin lymphoma (NHL), a malignancy that originates in B-cells, affecting different organs of the body. The Epstein-Barr virus-negative tumorigenic Ramos cell line was isolated from a 3-year-old Caucasian male patient with Burkitt’s lymphoma. Pretargeted radioimmunotherapy (PRIT) is a method of targeted delivery of high doses of radiotherapy to cancer cells. Yttrium-90 (90Y) and lutetium-177 (177Lu) are two beta-particle emitting radionuclides utilized for radioimmunotherapy. A 2015 study by Frost et al. published in PLoS One investigated the therapeutic potential of targeting 90Y or 177Lu to human B-cell lymphoma Ramos xenograft models in mice, using an anti-CD20 antibody-streptavidin conjugate (1F5-SA). Results showed therapy with 90Y-DOTA-biotin is significantly more efficient than with 177Lu-DOTA-biotin and could be advantageous for NHL patients. A 2006 Molecular Cancer Therapy article (Naumovski et al.) also used the Ramos model, in this case to study the anticancer effects of novel sapphyrin componds, which are all metal-free pentapyrrolic porphyrins. The group tested four derivatives, PCI-2050, PCI-2052, PCI-2051, and PCI-2053, and used intrinsic fluorescence to monitor biodistribution. Cytotoxicity analysis revealed that PCI-2050 is the most effective against tumor cells due to intrinsic propaoptotic activity and ability to localize to tumors. Finally, the 2008 Blood journal article (Gopal et al.) used the Ramos model to show that rituximab, an anti-CD20 drug with long in vivo persistence, circulating in xenograft models impairs CD20 directed radioimmunotherapy. This suggests that other lymphoma-associated antigens, such as CD45, should be used in combination therapy with rituximab. The Ramos cell line (human lymphoma; Burkitt’s lymphoma) is used to create the CDX (Cell Line Derived Xenograft) Ramos xenograft mouse model. The Ramos xenograft model is utilized to study antibody mediated therapeutic delivery (e.g. fusion protein L19-IL2, anti-CD37) and B-cell lymphoma sensitivity to TGF-β signaling (e.g. rituximab).
Download Altogen Labs Ramos Xenograft Model PowerPoint Presentation: ![]()
Basic study design
- All flasks containing Ramos cells are grown using RPMI-1640 medium supplemented with 10% FBS. Ramos cells are collected for injection and viable cells are determined (trypan blue exclusion). Cell concentration is diluted down to the density needed for inoculation.
- All study mice (athymic BALB/C or NOD/SCID) receive subcutaneous injections (s.c.) into a flank of one hind leg. Each injection contains 1 x 106 cells in a volume of 100 – 200 µL (Matrigel + Ramos cells suspension).
- Injection sites are palpated up to three times a week. When tumors are established, they are measured with calipers until they reach average sizes of 50-150 mm3. Treatment cohorts are formed by randomizing animals into necessary groupings. Injection of test materials are performed following the supplied treatment schedule.
- Tumors are tracked, measured daily and body weights recorded. Animals are humanely euthanized as tumor size reaches 2,000 mm3 (or size limits per approved IACUC protocol).
- Necropsy and tissue collections are performed, including tumors weighed and documented (optional: in vivo imaging).
- Standard gross necropsies enable tissue collection for downstream analysis. Collected tissues/tumors can be frozen, stabilized or nucleic acids isolated.
Get Instant Quote for
Ramos Xenograft Model
Xenograft animal models are used to assess the effectiveness of drugs against specific types of cancer. New medicines are tested on staged tumor growths that have been engrafted via subcutaneous or orthotopic inoculation in an immunocompromised mouse or rat model. All clinically approved anti-cancer agents have been evaluated with conventional preclinical in vivo models. Xenograft studies can be highly complex, starting with the selection of the appropriate animal model, choice of tumorigenic cell line, administration method, dosing, analysis of tumor growth rates and tumor analysis (histology, mRNA and protein expression levels).
Altogen Labs provides an array of laboratory services using over 90 standard CDX models and over 30 PDX models. Altogen Labs provides quantitative gene expression analysis of mRNA expression (RT-PCR) and protein expression analysis using the WES system (ProteinSimple). Animal handling and maintenance at the Altogen Labs facility is IACUC regulated. Following acclimation to the vivarium environment, mice are sorted according to body mass. The animals are examined daily for tumor appearance and clinical signs. We provide detailed experimental procedures, health reports and data (all-inclusive report is provided to the client that includes methods, results, discussion and raw data along with statistical analysis). Additional services available include collection of tissue, histology, isolation of total protein or RNA and analysis of gene expression.
Following options are available for the Ramos xenograft model:
- Ramos Tumor Growth Delay (TGD; latency)
- Ramos Tumor Growth Inhibition (TGI)
- Dosing frequency and duration of dose administration
- Ramos tumor immunohistochemistry
- Alternative cell engraftment sites (orthotopic transplantation, tail vein injection and left ventricular injection for metastasis studies, injection into the mammary fat pad, intraperitoneal injection)
- Blood chemistry analysis
- Toxicity and survival (optional: performing a broad health observation program)
- Gross necropsies and histopathology
- Lipid distribution and metabolic assays
- Imaging studies: Fluorescence-based whole body imaging, MRI
