Altogen Labs offers ‘Human –in- mouse’ xenograft (i.e. Patient derived xenografts, PDX) services for drug development of anticancer therapeutics [Full Article, PDF (240 kb)]
Slow progress in the oncology field is significantly attributed to the lack of faithful preclinical models exhibiting robust, predictive power in clinical trials. Patient derived tumor xenograft models (PDX models) are established to overcome most of these shortcomings by directly engrafting patient tumors in immune-compromised mice. These preclinical models have significant utility in translational research as they mimic patient tumor heterogeneity and have more predictive power than traditional methods. Many cancer-specific PDX models have been generated over the past years by many groups and are stable, retain principle histological parent tumor features, pathogenesis, global gene expression and therapeutic response. This review discusses different cancer specific PDX models, their establishment, achievements and limitations in terms of drug discovery, biomarker identifications, along with Altogen Labs PDX protocols and a discussion of limitations and future opportunities in the field.
The use of preclinical models for drug testing is central to translational cancer therapeutics. While mono-cellular layers of tumors cultivated in vitro and the mouse xenografts derived from those cells serve as useful screening tools, they often fail to recapitulate the key aspects of human malignancies and, thus, do not accurately predict drug efficacy in the clinical setting. Studies show mouse xenografts that have been selected and properly characterized have clear utility for predicting responsiveness to targeted agents. However, they do not replicate tumor microenvironments and interactions between the tumor and the innate immune system. The imperative for improved and more clinically predictive models for human cancer is the dire need of the hour (1, 2, 3).
Tumor graft models, i.e. Patient Derived Xenografts (PDX), are created by implanting primary human tumor materials directly into an immuno-deficient host (laboratory rats and mice). The mice used must be immuno-compromised to prevent transplant rejection. Several types of immuno-deficient hosts are used to establish PDX models, such as nude or SCID mice. The NOD-SCID mouse is considered more immuno-deficient than the nude mouse as they lack Natural Killer (NK) cells and, therefore, is more commonly used for PDX models. These models have the advantage over Cell Line Derived Xenografts (CDX models) as the tumor retains a more natural architecture and are more reflective of heterogeneity and histology as seen in primary tumors (2).
| Number | Characterstics | Cell Line In vitro | Cell Line Xenograft | PDX | Patient |
| 1. | Heterogeneity | Nil | Limited | Higher intra tumor heterogeneity | High intra tumor heterogeneity |
| 2. | Molecular sub types | Modest diversity | Modest diversity | Diverse range of molecular subtypes | Full range of molecular subtypes |
| 3. | Stroma | Nil | Murine stroma/ No human stroma | Mixed murine & human stroma | Human stroma |
| 4. | Growth Rate | Rapid | Rapid | Slow | Chronic |
| 5. | Stages | Mixed primary and metastatic | Mixed primary and metastatic | Mixed primary and metastatic | Predominately metastatic |
| 6. | Immune system | Nil | Limited | Severely Limited | Fully active |
| 7. | Clinical outcomes | Nil | Nil | Limited | Available |
Comparison of the PDX with previously established cell-lines, cell line xenografts, and donor tumor from which they are derived
Methodology
To establish a PDX model, patient tumors are obtained fresh from surgery, mechanically sectioned into fragments or chemically digested or physically manipulated into single-cell suspensions. They are then injected into NOD-SCID mouse. Use of tumor fragments retains cell-cell interactions and mimics the microenvironment while single cell suspensions enable unbiased sampling of whole tumor by eliminating segregates of sub clones. But cell viability and engraftment success are lower in single cell method compared to the tumor fragment method.
Tumors can be injected heterotopically or orthotopically. Heterotopic models involve injecting tumors in to subcutaneous flank of host. This provides easier cell transfer and precise monitoring of the growth and location. Orthotopic model involves direct implanting of tumor on an organ of choice. It is more technically challenging and time-consuming. In some cases in vivo imaging is required to verify tumor grafts after implantation. But they accurately mimic the human tumors from which they are derived in histology and gene expression. The generation harboring the patient-derived material is termed F0, with subsequent generations numbered consecutively (F1, F2, F3 and so on). It takes about 2 to 4 months for the tumor engraftment depending on the tumor type, implant location, and mice strain utilized. The engraftment failure should not be decided until at least 6 months (3). The model can be validated by comparing histopathologic, biologic and genetic features to its donor tumors. Studies have shown that these models retain donor tumor characteristics and these traits are maintained through successive mouse generations (4).
For drug development studies, expansion of mice after the F3 generation is often utilized (3, 5). Multilayered Biological assays like drug efficacy studies, combination studies and development of predictive biomarkers for novel therapies are performed on early generations (F3:F5), after ensuring that the PDX has not genetically or histologically diverged from the patient’s tumor.
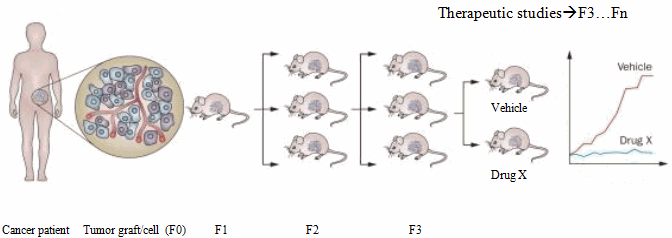
Establishment and testing of PDX models

Steps involved in PDX in vivo drug studies
PDX models are capable of maintaining the human malignancy complexity remarkably well. Hence they have extraordinary utility in basic research and demonstrated predictive power in novel drug efficacy studies, biomarker analysis and individualized patient studies (5,6). Thus the use of personalized tumor graft models can be powerful platform for therapeutic decision making and to efficiently guide cancer treatment in clinic. In conclusion, the evolution of our understanding of genome, the availability of genome wide screening, other technological advances like gene testing (BRCA1), biomarker identification (PSA), tissue monitoring (colonoscopy) and broader application of PDX preclinical models as tissue amplifiers, may lead us more effectively towards finding the designer drug/treatment for cancers.
Patient Derived Xenograft Models for Pancreatic Cancer
There are few therapeutic options available for pancreatic patients and new insights into pre-clinical models for therapeutic interventions are urgently needed. Ductal adenocarcinoma (PDA) is the most common form. Other forms include acinar cell carcinoma, pancreatic blastoma, and solid pseudopapillary neoplasm, all of them genetically distinct from PDA (7). Extensive genetic studies have shown that the key to understanding this complex disease lies in a core set of signaling pathways and processes. Whole-exome sequence analysis of primary tumors elucidated a core set of genetic pathways altered in this disease in association with an average of 63 genetic aberrations occurring within an individual tumor (8). But this extensive genetic information has not yet translated into improved clinical outcomes. Novel strategies are the need of the hour not only to develop new drugs, but also detect biomarkers that predict the efficacy of these drugs.
PDX models have so far shown to be stable and accurately reflect the tumor with regards to histopathology, gene expression, genetic mutations and therapeutic response. Several PDX models have been established and used in targeted therapy. In case of PDA, initially, studies were focused on developing, and characterize major drug-development applications which were further complicated by significant tumor heterogeneity both in gene and protein profiles (9, 10, 11). For example, these models predicted patients with high baseline expression of phosphorylated p60 S6 kinase would respond to mTOR inhibition which did not translate to patients. This may be due to low stringency in response to PDX models or multiple feedback loops in mTOR pathway (12). Another method is the use of PDX tumor biopsy for ex vivo therapeutic treatment and pharmacodynamics analysis for drug response and biomarkers for such responses. For E.g. Polo-Like Kinase 1 as a mediator of Gemcitabine resistance while Cyclin B1 as the biomarker of the response (13). Novel approach is the personalized drug therapy along with genome analysis. Studies treated PDX models with a panel of drugs while the patient receives the first line therapy. Drug demonstrating the most reactivity in PDX model is later administered as the cancer progresses. Whole genome analysis has equipped this strategy more specificity by identifying patient-specific mutations and correlation to PDX response to therapeutic agents (14). Garrito-Laguna et al revealed that tumor engraftment in PDX model correlates with poor survival in patients. They hypothesized it might be due to the loss of smad4 resulting in aggressive metastasis. Moreover, they also showed that stromal pathways are enriched in gemicitabine-resistant patients (15). In PDA, PDX models have shown that stromal modulation can increase intratumoral gemcitabine concentration to improve therapy efficacy (10,16). Importantly, gemcitabine and nabpaclitaxel have been shown to decrease intratumoral desmoplastic reaction leading to increased drug concentration and anticancer properties compared to individual drugs in PDX models of advance stage PDA in phase I-II clinical trials. Of the 44 patients treated a median survival increased to 12.2 months suggesting that PDX models can enhance our understanding of pancreatic cancer and its treatment.
Establishment of pancreatic xenograft model by Altogen Labs
PDX pancreatic models are established by the implantation of primary human PDA specimens either heterotopically or orthotopically. Orthotopic PDX models retain a greater proportion of stromal components and develop regional and distant metastases (9, 10). Here we are explaining the general protocol used in the establishing pancreatic PDX model in our company. All animal maintenance and procedures are conducted under institutional guidelines. Excess tissues from resected pancreatic carcinomas are generally used as the starting material. They are washed 3 times in antibiotic containing sterile PBS (50 units/ml penicillin and 50 ug/ml streptomycin) to reduce the contaminant load. Once washed, they are cut into 2-3 mm3 pieces with sterile blade in antibiotic containing PBS. It is important to make sure that tumor remains immersed in media all the time during the experiment. Non-necrotic pieces are then selected and immersed in matrigel.
In case of single cell suspensions of cancer cells instead tumor fragments, the tumor sample after the antibiotic wash is kept in collagenase IV (200 units/mL) solution. Using sterile blade, the fragments are minced into smallest possible fragment about 1 mm3 and about 0.5-0.6 g of the tumor is placed into each well of a 6-well plate. 5ml of collagenase IV solution is added to each well and pipetted thoroughly. The plate is incubated at 37oC for 2-3 h and pipetted every 20 min to increase the digestion. After digestion, serum free RPMI media is added and the solution is strained through a cell strainer. The cell solution is spun down at1200rpm for 5min. RBC lysis buffer is added and incubated for 10min. The resultant solution is spun down and washed twice in serum free RPMI media. The cell count is calculated using hemocytometer. About 500,000 to 1×106 cells/ 100ml Hank’s balanced salt solution containing 1% matrigel are generally used to inject. Immuno-deficient SCID mice of about 5-6 weeks age is generally used for the implantation. Under general anesthesia, an incision of about 2-3mm is made beneath the skin (subcutaneous) on either side of lower back or foot pad of the animal for heterotropic tumor formations. Tumors pieces (1 each) are implanted in these incisions. In case of single cells, suspensions 500,000 cells/100 ml Hank’s balanced salt solutions are injected into the desired implantation site. Orthotropic tumor formation involves direct implantation of these tumor fragments or direct injection of cell suspension into the pancreas during abdominal surgery in mice.
Tumors sizes are measured using digital calipers every 4 days and are allowed to grow till they are between 1,200-1,500 mm3. Tumors are then harvested after animal sacrifice and a portion is cryopreserved for biological assays. They are also passaged from parent generation (F1) to next generation thereby establishing a new patient derived xenograft model which can be used for drug studies or biomarker studies (F3…Fn). Tumors from F3 generation onwards are allowed to grow till 150-200 mm3 and randomly subdivided into different groups with 6 mice per group for various treatments (Eg. control, test article1, test article 2, test article n).
| Group | #Mice | Test article | Total volume | Route | Dosage |
| 1 | 20F | No Injection | n/a | n/a | n/a |
| 2 | 20F | Saline/Vehicle | 100 ul | I.P | n/a |
| 3 | 20F | Gemcitabine | 100 ul | I.P | 100 mg/kg/twice a week |
| 4 | 20F | Gemcitabine | 100 ul | I.P | 50 mg/kg/twice a week |
| 5 | 20F | Erlotinib | 100 ul | I.P | 100 mg/kg/day |
| 6 | 20F | Erlotinib | 100 ul | I.P | 50 mg/kg/day |
Standard drug study in pancreatic cancer patient derived xenograft model
The experiments are usually terminated between 4 weeks to 14 weeks depending on the study requirements. The mice are monitored daily for toxicity, weighed thrice a week and tumor size is measured by caliper methods. Standard formula used: tumor volume= (length-width)/2. Relative tumor growth inhibition= relative tumor growth in treated mice/ relative tumor growth in control (T/C).
Prostate cancer patient derived tumor xenograft models.
Prostate cancer is the second most commonly diagnosed cancer in men and one of the leading causes of death (17). As prostate cancer in general is androgen-dependent, androgen ablation therapy of locally advanced forms are effective. But tumor returns within 18 to 24 months as incurable aggressive androgen independent, castrate-resistant form (CRPC). It has been shown that emergence of CRPC manifests as rising serum Prostate Specific Antigen (PSA) which in turn is mediated by androgen receptors (AR) (18). Several cell migration and invasion pathways like PI3K/AKT pathways and multiple gene rearrangements have been shown to play a major role in the progression of prostate cancer (19). Therapies targeting the AR and other pathways can improve the survival rate quality of life in advanced prostate cancer patients. Many costly therapeutic clinical trials have failed to improve patient outcomes. These failures are mainly due to the high clinical and biologic tumor heterogeneity, lack of distinguishable histologic subtypes and hence a lack of reliable preclinical models (20). Patient derived tumor xenograft models along with next generation sequencing and integrated genomics seems to be the tools of future in understanding the progression of prostate cancer to CRPC as well as the development of effective therapies. Several studies have established PDX models and reported different prostate cancer specific biological processes like the transition of adenocarcinoma to neuroendocrine carcinoma during and after androgen ablation therapies (21), or hormonal carcinogenesis involving the epithelial and stromal interactions (22), or integrated genome and transcriptome analysis to understand the mechanism of aggressive forms (23).
Establishment of Prostate xenograft model by Altogen Labs
PDX prostate models are established by the implantation of primary human tumor specimens either heterotopically or orthotopically. The tumor engraftment can be done subcutaneous, subcapsular renal and prostatic orthotopic sites with most efficient engraftment being subcapsular renal (>90%). They also mimic tumor microenvironment including continued expression of androgen receptor and PSA levels (24). Here we are explaining the general protocol used to establish prostate cancer PDX model in our company. All animal maintenance and procedures are conducted under institutional guidelines.
Prostate cancer cell or tissues are extremely difficult to grow in labs and all models including patient derived xenograft models are from patients with advanced forms and with modifications like including of fibroblasts (25). Transurethral sections from patient with prostate cancer are received immediately after surgery in sterile PBS. A portion of the tissue is cryopreserved for future analysis. They are washed 3 times in antibiotic containing sterile PBS (50 units/ml penicillin and 50 ug/ml streptomycin) to reduce the contaminant load. Once washed, they are cut into 1-3 mm3 fragments with sterile blades. In case of heterotopic xenografting fragment no greater then 1 mm3 is implanted subcutaneously, bilateral through small incisions above the scapula in male SCID mice. Another option is the implantation of tumor fragment on the sub-renal capsule (SRC) sites of the mice.
Under general anesthesia, an incision in made about 2cm along the dorsal midline of the mouse. On the body wall incisions shorter than the long axis of the kidney is made and kidney is gently pushed out of the body cavity. Then the capsule is exposed using forceps, and a cavity is made between capsule and parenchyma. About 2-3 grafts per kidney are transferred to the cavity using fire-polished glass pipette. The left kidney is easier to access because of a more caudal location in the abdominal cavity. If bilateral transplantation is required, the same procedure is repeated for right kidney. In case of orthotopic xenografts, about 2 mm3 tumor fragments are implanted on to the capsule of the anterior prostate between the two prostate ducts under general anesthesia. Generally, mice 5-6 weeks old are used for the xenograft models. They are previously supplemented with 25 mg testosterone via 1 cm implanted testosterone pellets to augment the tumor growth. Plasma testosterone levels in tumor bearing mice with implants are monitored by radioimmunoassay.
Tumors sizes are measured using digital calipers every 4 days and are allowed to grow till they are between 5-10 mm in diameter. Tumors are then harvested after animal sacrifice and a portion is cryopreserved for biological assays. They are also passaged from parent generation (F1) to next generation by serial transplantation there by establishing a new patient derived xenograft models. Tumors from F3 generation onwards are allowed to grow till 30-50 mm3 and randomly subdivided into different groups of treatment with 6 mice per group including control, test article1, test article 2, test article n etc.
| Group | #Mice | Test article | Total volume | Route | Dosage |
| 1 | 20M | No Injection | n/a | n/a | n/a |
| 2 | 20M | Saline/Vehicle | 50 ul | I.P | n/a |
| 3 | 20M | Docetaxel | 50 ul | I.P | 5 mg/kg/twice a week |
| 4 | 20M | Docetaxel | 50 ul | I.P | 50 mg/kg/twice a week |
| 5 | 20M | Aneustat | 50 ul | I.P | 100 mg/kg/twice a week |
| 6 | 20M | Aneustat | 50 ul | I.P | 1000 mg/kg/twice a week |
Standard drug study in prostate cancer patient derived xenograft model
The experiments are usually terminated between 4 weeks to 14 weeks depending on the study requirements. The mice are monitored daily for toxicity, weighed thrice a week and tumor size is measured by caliper methods. Tumor volume is calculated from two tumor diameter at right angle using the following formula V= P/6(d1.d2)3/2 (volume of an ellipse). Relative tumor growth inhibition= relative tumor growth in treated mice/ relative tumor growth in control (T/C).
Colorectal Cancer (CRC) patient derived tumor xenograft models
Colorectal cancer (CRC) is the third common malignancy in the world with approximately 1 million new cases reported every year. The development of CRC is a multistep process in which accumulation of many genetic/epigenetic alterations leads to the progression of normal intestinal epithelial cells to dysplastic tissue to benign adenoma and finally to full blown metastatic carcinoma. Alterations in Wnt pathway and/or inactivation of APC gene initially drive this process, while activations of Ras-MAPK pathway or TFG-β receptors transform benign adenomas to metastatic cancer. Thus this complex condition involves of several pathways and epigenetic or post translational modifications making it highly challenging to find clinical favorable therapeutic targets (12,23). Despite the improvements in the systemic therapy of CRC over the years, almost half of the patients undergoing surgical resection along with chemotherapy, experience relapse and metastasis occur in about 50% of the patients. Lack of understanding of the molecular nature of metastasis along with the existence of a few biomarkers for early detection and therapy resistance makes successful treatment a myth (26). Long-term survival of CRC patients is correlated with disease stage at diagnosis, and the 5-year survival rate for patients with metastatic CRC is less than 10%. Newer targeted agents like panitumumab or monoclonal antibodies like Trastuzumab and Ramucirumab targeting molecular pathways are used in treating metastatic CRC, but some patients do not respond to these targeted therapies indicating the need to develop personalized drug treatment for patients (18).
CRC PDX models are easier to establish with an intake rate of 75% in mice. Even early stage tumor exhibits chromosomal instability, hence establishment and propagation of PDX models retain the chromosomal abnormalities, intratumoral clonal heterogeneity and histology of the parent tumor. Thus establishing PDX models and mutation analysis/gene profiling using NGS is the future for better clinical outcomes by targeted therapies in CRC. Studies have shown that CRC PDX model responds to standard anticancer drugs like 5-flurouracil and the response correlates with the response in patients under these drugs. Moreover they also retained the histological features of the parent tumor though there was an elevated level of biomarkers like CEA (Carcino Embryonic Antigen). Another aspect of anti-cancer therapy focuses on the mechanism of resistance to standard agents that most of the patients develop and determine a rational combination strategy to overcome this. Cetuximab (Erbitux) is an approved agent for CRC that targets epidermal growth factor receptor (EGFR). In spite of the receptors wide distribution only a subgroup of patients respond to cetuximab. Studies on PDX models from CRC patients have shown that downstream pathways involving MET, KRAS, NRAS and BRAF plays an independent role in developing this resistance. A combination therapy involving cetuximab and MET inhibitors were addictive and hence could be of high clinical significance. PDX models were also able to functionally cross validate many biomarkers like HER2; emphasizing that genomic analysis along with PDX models can bring about revolutionary changes in the oncology field (27).
Establishment of CRC PDX models by Altogen Labs
Here we are explaining a general protocol used in the PDX model establishment in our company. All animal maintenance and procedures are conducted under institutional guidelines. Excess GC tissues from carcinomas are generally used as the starting material. They are washed 3 times in antibiotic (50 units/ml penicillin and 50 ug/ml streptomycin) containing sterile PBS to reduce the contaminant load. Once washed, they are cut into 1-2 mm3 pieces with sterile blade in antibiotic containing sterile PBS and immersed in matrigel. Six to eight weeks old SCID mice are generally used for human colorectal tissue implantation.
Each mouse is treated with analgesic ketoprofen (5 mg/kg body weight) with betadine being used to sterilize the right flank before surgery. Under general anesthesia, matrigel embedded fragments are inserted into subcutaneous pockets on the lower back or scapular region for heterotropic engraftment. In case of orthotropic implantation, tumor fragments are attached to the cecum to be entirely surrounded by serosa of intestine. Meanwhile, representative portions of fresh parent tumor are fixed in 10% formalin buffer for 24 hours and paraffin-embedded for pathological assessment. Postoperative cares include daily animal monitoring for overall health and tumor growth. Tumor growth is monitored and measured for 12-14 weeks.
Tumors sizes are measured using digital calipers every 4 day and tumor volume is calculated as follows: V = π/6 × length × width × width. Tumors are allowed to grow till they are between 1,500 – 2,000 mm3 and then harvested after animal sacrifice. A portion is again cryopreserved for biological assays. They are also passaged from parent generation (F1) to next generation thereby establishing new patient derived xenograft model. Tumors from F3 generation onwards are allowed to grow till 100-400 mm3 and randomly subdivided into different groups of treatment with 6 mice per group including control, test article1, test article 2, test article n etc
| Group | #Mice | Test article | Total volume | Route | Dosage |
| 1 | 25F | No Injection | n/a | n/a | n/a |
| 2 | 25F | Saline/Vehicle | 100 ul | I.P | n/a |
| 3 | 25F | Ceturimab | 100 ul | I.P | 100 mg/kg/twice a week |
| 4 | 25F | Ceturimab | 100 ul | I.P | 50 mg/kg/twice a week |
| 5 | 25F | Bevacizumab | 100 ul | I.P | 25 mg/kg/day |
| 6 | 25F | Bevacizumab | 100 ul | I.P | 50 mg/kg/day |
Standard drug study in CRC cancer patient derived xenograft model
The experiments are usually terminated between 4 weeks to 14 weeks depending on the study requirements. Tumor volume is calculated from two tumor diameter at right angle using the following formula V= P/6(d1.d2)3/2 (volume of an ellipse). Relative tumor growth inhibition= relative tumor growth in treated mice/ relative tumor growth in control (T/C).
Breast Cancer PDX models
Breast cancer is one of the most frequently diagnosed types of cancer in women and standard care for the patients is Neoadjuvant chemotherapy (NeoCT). But 10-30% patients do not respond to the therapy and relapse rates are high. So there is a pressing need to understand the mechanism of resistance as well identify novel therapeutics and biomarkers in those patients (28). Breast Cancer is a diverse and complex disease involving distinct molecular types, unique gene-expression profile, which might influence the responses to standard or novel therapies. These molecular classifications: luminal A, luminal B, triple negative (lack the expression of estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor 2 (HER-2)), basal-like and HER-2 positive: provide a unique platform to develop patient-specific therapies depending on tumor subtypes. The intratumoral heterogeneity of breast cancer is the major stumbling block in developing patient derived xenograft models. Recent large scale interest in developing PDX models for such hormone driven cancer models has led to the emergence of PDX models has major tool for translational research with the promise of developing a more personalized patient therapy.
Multiple groups have now established PDX models with a bias towards triple negative forms due to their aggressive engraftments and growth. ER-Positive tumors are very difficult to engraft and shows Luminal B rather than A enrichment using Ki67 staining. Basal-like tumors are typically triple negative and carry the worse prognosis due to relapse. Systemic treatment is limited to cytotoxic chemotherapy, thus highlighting the need for biomarker development. PDX model studies paved the way of new classes of treatments including inhibition of PI3K pathway, NOTCH signaling, checkpoint kinase1 pathway, or Wnt pathway (13,25). TNBC shows a high degree of chromosomal rearrangements suggesting DNA damaging agents might be highly efficient clinically (23). Also PDX models of TNBC have shown that there is an elevated level of BCL-2 suggesting another potential biomarker for the prognosis. ER-positive PDX models are estrogen dependent and respond to hormone therapy similar to clinical response. Valliant et al in 2013 showed that targeting BCL-2 was essential for tumor growth and there is a synergistic interaction between BCL-2 inhibition, tamoxifen and mTOR inhibition suggesting novel estrogen dependent signaling pathways. Very few studies have been carried out with HER-2 positive PDX models. There are two therapeutic strategies used in HER-2 positive tumor patients: first monoclonal antibodies targeting the receptor (Trastuzumab , Pertuzumab) second is ATP competitors that inhibit the phosphorylation of HER-2 intracellular kinase domain (Lapatinib, Neratinib). Marangoni et al 2007 showed a synergistic interaction in HER-2 PDX models while targeted with trastuzumab and docetaxel suggesting a better anti-tumor therapy.
Establishment of breast cancer patient derived tumor xenograft models by Altogen Labs.
Briefly, for initial transplantation tumors derived from primary surgical resection are sliced into fragments and then implanted into immune-compromised SCID mice. In other cases, cell suspensions consisting of pleural or peritoneal fluid is injected into the SCID mice. Usually, breast tumors are implanted subcutaneous or hind leg fat pad or scapular fat pad (heterotropic), and orthotopic implantation involve inguinal mammary fat pad because it mimics the tumor stromal microenvironment and develops better regional and distant metastases (9). Here we are explaining a general protocol used in the PDX model establishment in our company. All animal maintenance and procedures are conducted under institutional guidelines.
The tumor samples from surgery are placed in sterile PBS containing antibiotics and brought immediately within 1h to animal facility. Portions are frozen or paraffin embedded for future analysis. Rest of the tumor samples is washed 3 times in antibiotic containing PBS to reduce the contaminant load. Once washed, they are cut into 1-3mm3 pieces with sterile blade in antibiotic containing PBS and immersed in 1% matrigel. Four-six weeks old female SCID mice are anesthetized, and analgesia is administrated with carprofen (5 mg/kg) subcutaneously. Their hair is clipped and skin is sterilized with povidone-iodine and alcohol. A small incision is made in the lateral flank and tumor chucks immersed in matrigel is implanted into the mammary fat pad of mice (in case of orthotopic xenografts). The tumor is inserted in the subcutaneously in the scapular region/hing leg for heterotropic implant. The hosts are also implanted with estradiol pellet (0.36 mg-0.5 mg/pellet/over 60days) for tumor growth. In case of single cell suspension transplantation, the tumors are minced and digested with 150 U/ml collagenase and 50U/ml hyaluronidase for1h at 37oC. The resulting suspension is again digested with 0.5% trypsin-EDTA and 5mg/ml dispase for 1h at 37oC. The solution is filtered and RBCs are removed by cell lysis (described earlier). About 150,000-250,000 cells are resuspended in 50% FBS containing matrigel solution is injected into the site of implantation.
Tumors sizes are measured using digital calipers every 4 days and are allowed to grow till they are between 600-1000 mm3. Tumors are then harvested after animal sacrifice and a portion is cryopreserved for biological assays. They are also passaged from parent generation (F1) to next generation thereby establishing a new patient derived xenograft model which can be used for drug studies or biomarker studies (F3…Fn). Tumors from F3 generation onwards are allowed to grow till 80-160 mm3 and randomly subdivided into different groups for drug studies with 6 mice per group including control, test article 1, test article 2, test article n etc.
| Group | #Mice | Test article | Total volume | Route | Dosage |
| 1 | 20F | No Injection | n/a | n/a | n/a |
| 2 | 20F | Saline/Vehicle | 100 ul | I.P | n/a |
| 3 | 20F | Cisplatin | 100 ul | I.P | 1 mg/kg/once a week |
| 4 | 20F | Cisplatin | 100 ul | I.P | 1 mg/kg/twice a week |
| 5 | 20F | Doxorubicin | 100 ul | I.P | 2 mg/kg/every 3 weeks |
| 6 | 20F | Doxorubicin | 100 ul | I.P | 2 mg/kg/week |
Standard drug study in breast cancer patient derived xenograft model
The experiments are usually terminated between 4 weeks to 14 weeks depending on the study requirements. The mice are monitored daily for toxicity, weighed thrice a week and tumor size is measured by caliper methods. Standard formula used: Tumor Volume = [Length – Width] divided by 2. Relative tumor growth inhibition = relative tumor growth in treated mice/ relative tumor growth in control (T/C).
Lung Cancer Patient derived Xenograft models
Lung cancer is the leading cause of cancer related death worldwide with about 1.4 million deaths per year. There are three main types of lung cancer. 1. Non-Small Cell Lung Cancer (NSLC) is the most common and accounts for about 85% of cases. Squamous cell carcinoma, adenocarcinoma and large cell carcinoma are the subtypes. 2. Small Cell Lung Cancer (SCLC) accounts for about10-15% of cases and the most aggressive form. 3. Lung carcinoid Tumor (lung neuroendocrine tumors) accounts for fewer than 5% of cases. They grow slowly and rarely spread (27). The survival rate of these patients has improved during the past decade with new strategies like platinum based adjuvant therapies, but the tumors nearly always recur. They are usually more aggressive and resistant to subsequent chemotherapy regimens (28). The treatment options available for the patients with inoperable solid tumors remains mostly unchanged compared to therapies offered decades ago. The main reason for the slow progress in the discovery and development of novel therapeutic agents is the lack of appropriate tumor models. The optimal model should enable preclinical studies for drugs, produce comparable response to standard drugs in vivo and should mimic human tumor biology and microenvironment (30). To overcome these hurdles, PDX models are established by implanting tumors fragments or cells, dissected out fresh from the patients, in immune-compromised mice. They preserve the genomic integrity and tumor heterogeneity in vivo. Thus PDX models faithfully recapitulate the tumor biology and exhibit similar chemo responsiveness to anti-neoplastic agents as observed in same donating patients in several cancer types as explained earlier in this review.
Since the approval of oral Crizotinib (ALK -Anaplastic Lymphoma Kinase and MET or HGFR- Hepatocyte Growth Factor Receptor inhibitor) and Erlotinib (EGFR- Epidermal Growth Factor Receptor inhibitor) for NSLC patient treatment, there is a renewed interest in PDXs, their mechanism of responsiveness and resistance to standard or novel agents. These agents show antitumor activities by the inhibition of many kinases pathways that in turn lead to inactivation of cell survival and proliferation pathways like mTOR pathways and PI3K/Akt pathways. This leads to cell arrest, apoptosis and finally cell death. Gene profiling of PDX models from SCLC patients showed a similar expression pattern as the donor and is maintained during the early serial passages thereby consolidating their clinical relevance. ABT 737 is an inhibitor for BCL2, a central regulator of cell survival. It has potent lethal effect on lung cancer cell lines and cell line induced xenograft. But Han et al in 2008 showed that SCLC PDX models show selective inhibition in tumor growth suggesting that ABT 737 might not be an ideal candidate for clinical trials for lung cancer. This data have specific implications for anti BCL2 drug developments as well as the importance of PDX as preclinical model in the developing novel anticancer drugs and biomarkers. Molecular cytogenetic analysis of NSLC PDX revealed complex chromosomal aberrations with multiple numerical and structural changes in the cancer cells with karyotypes often near triploid. The most common EFGR mutation associated with NSLC are small in-frame deletions in exon 19 (△LREA motif) and the L858R point mutation in exon 21.
Studies also showed that rate of tumor engraftment correlate with size of the tumor, poor differentiation, and K-RAS mutations in NSLC types. Also, the squamous carcinoma forms xenografts aggressively compared to adenocarcinoma. Thus the tumor engraft-ability is directly correlated to more aggressive clinical and biological pathology. This is an interesting correlation that can be clinically significant to predict patient survival. Further in the lines of correlation, Dong et al in 2010 showed PDX from NSLC patients had viable nest of cells even while responding to drugs highlighting the intratumoral heterogeneity in drug response. Secondly six of the seven PDX models did not respond to ex vivo chemotherapy, mimicking the donor patients. Zhang et al 2012 showed FGFR-1 amplification correlates with squamous lung cancer and this in turn correlates with smoking status of the patients. They were able to drive tumor regression in PDX models of FGFR-1 amplified squamous cell lung cancer using novel and potent FGFR inhibitor, AD4547. These studies further support the use of PDX models in developing novel, effective patient specific chemotherapy regimens.
Establishment of Lung PDX models by Altogen Labs
PDX lung models are established by the implantation of primary human lung tumor specimens either heterotopically or orthotopically. Here we are explaining the general protocol used in the establishing Lung cancer PDX model in our company. All animal maintenance and procedures are conducted under institutional guidelines. Excess tissues from resected lung carcinomas or tumor cells isolated from pleural effusion or bronchoscopic biopsy (single cell suspensions) are generally used. Tumors tissues are washed 3 times in antibiotic containing sterile PBS (50 units/ml penicillin and 50 ug/ml streptomycin) to reduce the contaminant load. Once washed, they are cut into 1-3 mm3 pieces with sterile blade in antibiotic containing PBS. They are carefully cleared of necrotic tissues and non-necrotic pieces are immersed in matrigel for implantation. The cell samples are obtained by endobronchial trans needle aspiration either from a primary tumor or suspected nodal tissue or pleural effusion are prepared in to single cells suspension (500,000 cells/100 ml Hank’s balanced salt solution containing 1% Matrigel solution) for implantation. Immunodeficient NOD/SCID mice of about 6-8 weeks age is generally used for the implantation.
Under general anesthesia, an incision of about 2-3mm is made beneath the skin (subcutaneous, scapular, subrenal capsule, mammary/ hind limb pads) for heterotropic tumor formations. Tumors pieces (1 each) are implanted in these incisions as explained earlier. In case of single cells suspensions 500,000 cells/100 ml Hank’s balanced salt solutions is injected into the desired implantation site. Orthotopic tumor formation involves direct implantation of these tumor fragments or direct injection of cell suspension into the lung during abdominal surgery in mice. Tumors sizes are measured using digital calipers every 4 days. Mice are sacrificed once the tumor grows about 1000-1200 mm3 and a portion is cryopreserved for biological assays. They are also passaged from parent generation (F1) to next generation thereby establishing a new patient derived xenograft model which can be used for drug studies or biomarker studies (F3…Fn). Tumors from F3 generation onwards are allowed to grow till 100-200mm3 and randomly subdivided into different groups with 6 mice per group for various treatments (Eg. control, test article1, test article 2, test article n).
| Group | #Mice | Test article | Total volume | Route | Dosage |
| 1 | 20F | No Injection | n/a | n/a | n/a |
| 2 | 20F | Saline/Vehicle | 100 ul | I.P | n/a |
| 3 | 20F | Erolotinib | 100 ul | I.P | 100 mg/kg/twice a week |
| 4 | 20F | Erolotinib | 100 ul | I.P | 50 mg/kg/twice a week |
| 5 | 20F | Test X | 100 ul | I.P | 100 mg/kg/day |
| 6 | 20F | Test X | 100 ul | I.P | 50 mg/kg/day |
Standard drug study in lung cancer patient derived xenograft model
The experiments are usually terminated between 4 weeks to 14 weeks depending on the study requirements. The mice are monitored daily for toxicity, weighed thrice a week and tumor size is measured by caliper methods as explained earlier.
PDX model offers a powerful tool in studying tumor biology, evaluating standard/novel anticancer drugs, developing biomarkers, genes for drug resistance and recurrences. They are arguably the most faithful model of parent tumor heterogeneity but have inherent challenges and potential opportunities. Although the concept and initial establishment of PDX models have been in existence for decades, their value in oncology drug development is just becoming realized as personalized medicine strategies transform cancer therapy. A notable example is pilot clinical study using pancreatic PDX models to guide treatment of 11 patients with advanced cancer. Seventeen treatment plans were devised with 15 of these resulting in durable partial remissions (22).
But PDX models have inherent limitations which cannot be ignored. One of the major drawbacks of PDX models is that human stroma is substitute by murine stroma throughout the tumor growth in mice. As they are passaged from generations to generations they are rapidly replaced by murine stroma (27). These aspects should be kept under consideration while using these models for drug testing/biomarker identification, given the importance of stromal microenvironment in numerous aspects of cancer biology and drug response. Another major shortcoming is the lack of immune system in the host mice. Tumors are highly antigenic, and patient’s immune system is on overdrive during tumorogenesis. These aspects cannot be predicated or studied in immune-deficient mice host. Traditionally drug screening/testing was done with cell lines but they have poor predictive value and more permissive as explained earlier. Using PDX model is ideal but it is highly time-consuming and is expensive to maintain such ‘live’ tumor banks (29). Since some PDX models are comparatively easier to establish like hormone derived tumor derived models, there is an over representation of such PDX models making it tougher develop personalized medicine for less established PDX varieties. Hence standardized international criteria should be established for PDX establishments, drug/biomarker screening and testing, response stringency with molecularly defined tumor subtypes, which can result in personalized medicine strategies for cancer patients.
There is a growing interest in establishing PDX models and their diverse applications but there is a need for additional research on diverse issues like implantation procedures including type, locations, tissue types, time lines, engraftment rates, new mouse stains, compensating the absence of full-fledged immune system, better elucidation of biomarkers and drug response as well as better/novel applications of these models. Future strategies in personalized medicine using PDX model include more sophisticated orthotopic models, in vivo functional siRNA transfection models and genomic screening, patient-matched reconstitution of robust immune system by co engraftment of bone marrow stem cells, and patient-matched stromal component engraftment (30). These humanized PDX models could allow researchers and clinicians to predict and explained more sophisticated tailor- made therapy for individual patients.
Mouse strains available at Altogen Labs:
| Mouse type | T cells | B cells | NK cells | Coat | Other Notes |
| CD1 | No | Yes | Yes | White/albino | Outbred |
| B6 | Yes | Yes | Yes | White/albino | Inbred |
| Balb/c | No | Yes | Yes | Nude, albino | Inbred |
| Balb/SCID | No | No | Yes | White | Inbred |
| NOD/SCID | No | No | Impaired | White | Inbred |
| Nu/nu | No | Yes | Yes | Nude | Outbred |
| CD57BL | Yes | Yes | Yes | Dark brown/black | Inbred |
| CB17 | Yes | Yes | Yes | White | Inbred |
| NSG | No | No | No | White | Inbred |
| Swiss Nude | No | Yes | Yes | Nude | Outbred |
About the models
CD1
This model originates from a non-inbred Swiss stock of the 1920s from the Centre Anticancerux Romand (Lausanne, Switzerland). Outbred stocks are generally used for their genetic variability.
B6
This strain of mouse arose from a spontaneous mutation in the C57BL/6 strain resulting in a coisogenic albino mutant. These mice have a mutant tyrosinase gene.
Balb/c
This strain of nude mouse was developed in the 1980s through many crosses and backcrosses and remains to be an inbred model. Balb/c mice do not have a thymus and therefore cannot produce T-cells and are considered immunodeficient. Balb/c mice are often used for their easy breeding and similar weights (low-variation) of males and females. They are also used for monoclonal antibody production.
Balb/SCID
This mouse model lacks functioning T and B cells but do have functioning NK cells which limits engraftment. These mice are sensitive to irradiation and have functioning macrophages, dendritic cells and complement activity. Some cancer cell lines show improved engraftment over nude models in Balb/SCID mice.
NOD/SCID
The homozygous SCID mutation results in impaired T cell and B cell lymphocyte development. The NOD characteristic results in impaired natural killer cell function. NOD/SCID mice also lack macrophage and dendritic cell activity as well as reduced complement activity. These mice have a non-obese diabetic and insulitis background and low cytokine production. NOD/SCID mice exhibit a 36-week median survival due to the development of thymic lymphomas, which limits their use to short-term experiments.
Nu/nu
These mice originate from the National Institute of Health (NIH). Originally thought to be BALB/C congenic mice, once it was discovered that these mice were outbred they were determined to be of their own strain. These mice do not have a thymus, or T-cells, and are nude immunodeficient models.
CD57BL/6
This laboratory mouse strain was the 2nd mammalian species to ever have its genome published in entirety. They originate from the Bussey Institute for Research in Applied Biology in 1921. These mice are often selected for easy breeding and availability of congenic strains. These mice are particularly sensitive to odors, noise, pain, cold, alcohol and morphine addiction.
CB17
CB17 mice are of a congenic strain that carry the immunoglobulin heavy chain allele (Igh-1b) from a C57BL/Ka on a BALB/c background. They are an ideal control for the CB17/SCID immunodeficient mouse model
NSG
Also known as NOD scid gamma, these mice are deficient in NK, T and B cells as well as multiple cytokine pathways. They also have reduced dendritic cell function and defective macrophage activity and lack a complement system. They are one of the most immunodeficient models available and unlike NOD/SCID mice, NSG mice do not develop thymic lymphomas and can be used for long-term experiments.
Swiss Nude
These mice originate from the 1974 Gustave Roussy Institute (Villejuif, France) Swiss stock. They are T cell deficient, nude and albino.
References
- Siolas D et al. (2013) Patient Derived Tumor Xenografts: transforming clinical samples into mouse models.
- Kopetz S et al. (2012) The Promise of Patient-Derived Xenografts: The Best Laid Plans of Man and Mice.
- Tentler J et al. (2012) Patient-derived tumour xenografts as models for oncology drug development.
- Morton C et al. (2007). Establishment of human tumor xenograft in immunificient mice.
- Hidalgo M et al. (2014). Patient-Derived Xenograft Models: An emerging platform for translational cancer research.
- Kim M et al. (2009). Generation of orthotopic and heterotopic human pancreatic cancer xenografts in immunodeficient mice.
- Yeo T et al. (2002). Pancreatic Cancer.
- Rubio-Viqueira B et al. (2009). Direct in vivo xenograft tumor model for predicting chemotherapeutic drug response in cancer patients.
- Jones S et al. (2008). Core signaling pathways in Human Pancreatic Cancers revealed by global genomic analysis.
- Fu X et al. (1992). A metastatic nude-mouse model of human pancreatic cancer constructed orthotopically with histologically intact patient specimens.
- Rubio-Viqueira B et al. (2006). An in vivo platform for translational drug development in pancreatic cancer.
- Laguna et al. (2010). Integrated preclinical and clinical development of mTOR inhibitors in pancreatic cancer.
- Jimeno A et al. (2010). A fine needle aspirate based vulnerability assay identifies polo-like kinase 1 as a mediator of Gemcitabine resistance in pancreatic cancer.
- Villarroel M et al. (2011). Personalizing cancer treatment in the age of global genomic analyses: PALB2 gene mutations and the response to DNA damaging agents in pancreatic cancer.
- Olive K et al. (2009). Inhibition of Hedgehog signaling enhances delivery of chemotherapy in a mouse model of pancreatic cancer.
- Siegel R et al. (2012). Cancer statistics.
- Qu S et al. (2014). Enhanced anticancer activity of a combination of docetaxel and Aneustat (OMN54) in patient derived advanced prostate cancer tissue xenograft model.
- Lin D et al. (2014). High fidelity patient derived xenografts for accelerating prostate cancer discovery and drug development.
- Yoshida T et al. (2005). Antiandrogen bicaluamide promotes tumor growth in a novel androgen dependent prostate cancer xenograft model derived from a bicaluamide treated patient.
- Young M et al. (2013). What are the best routes to effectively model human colorectal cancer?
- Oh B et al. (2015). Correlation between tumor engraftment in patient-derived xenograft models and clinical outcomes in colorectal cancer patients.
- Chiron M et al. (2014). Differential Antitumor Activity of Aflibercept and Bevacizumab in Patient-Derived Xenograft Models of Colorectal Cancer.
- Fichtner I et al. (2004). Anticancer drug response and expression of molecular markers in early-passage xenotransplanted colon carcinomas.
- Bertotti A et al. (2011). A molecularly annotated platform of patient-derived xenografts (‘xenopatients’) identifies HER2 as an effective therapeutic target in cetuximab-resistant colorectal cancer.
- DeRose Y et al. (2011). Tumor grafts derived from women with breast cancer authentically reflect tumor pathology, growth, metastasis and disease outcomes.
- Lehmann B et al. (2014), PIK3CA mutations in androgen receptor-positive triple negative breast cancer confer senstitivity to the combination of PI3K and androgen receptor inhibitors.
- Scott A et al. (2013). Preclinical and clinical studies of gamma secretase inhibitors with docetaxel on human breast tumors.
- McAuliffe P et al. (2015). Ability to generate patient derived breast cancer xenografts is enhanced in chemoresistant disease and predicts poor patient outcomes.
- Whittle J et al. (2015). Patient derived xenograft models of breast cancer and their predictive power.
- Aparicio S et al. (2015). Examining the unitlity of patient derived xenograft mouse models.
