The HCC827 cell line is characterized by its epithelial morphology and is widely used as an in vitro model for studying the molecular mechanisms driving lung cancer. HCC827 cells harbor an EGFR exon 19 deletion mutation, making them particularly sensitive to EGFR-targeted therapies, and they exhibit high levels of EGFR expression. The cell line is commonly utilized in drug screening, resistance studies, and therapeutic cancer research, offering valuable insights into the development of targeted treatments for NSCLC. Furthermore, HCC827 cells are frequently employed in preclinical studies to explore EGFR signaling pathways, tumor progression, and potential therapeutic agents. Their strong growth characteristics and clinical relevance make HCC827 cells a crucial resource for advancing lung cancer research. Lung cancer remains one of the leading causes of cancer-related deaths worldwide, and despite advancements in early detection and treatment, the prognosis for NSCLC patients remains poor, primarily due to late-stage diagnoses and the development of resistance to therapies. Xenograft models, in which human cancer cells are implanted into immunocompromised mice, have become invaluable tools in the study of NSCLC. These models allow for the evaluation of tumor growth, metastasis, and response to novel therapeutic agents in a more biologically relevant context. Specifically, NSCLC xenografts provide a platform to investigate the molecular underpinnings of the disease, including the role of key mutations and signaling pathways that drive tumorigenesis. As these models closely replicate human disease, they are critical for preclinical testing of potential treatments and for developing personalized cancer therapies. Consequently, NSCLC xenografts are essential in advancing the understanding and treatment of lung cancer.
HCC827 Lung Cancer Subcutaneous and Metastatic Model: Download ![]()
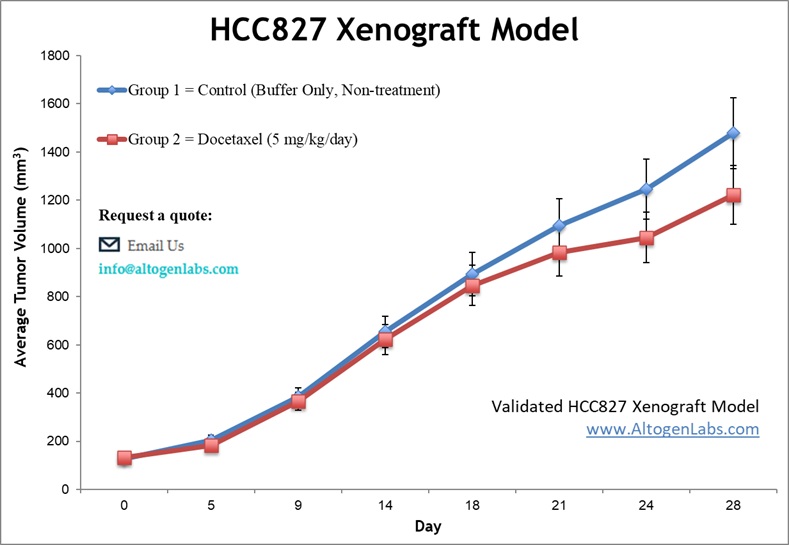
The HCC827 xenograft model offers a valuable platform for preclinical studies aimed at evaluating therapeutic agents targeting EGFR-driven lung adenocarcinoma. At Altogen Labs, researchers examine key experimental endpoints, such as Tumor Growth Delay (TGD) and Tumor Growth Inhibition (TGI), which can provide crucial insights into the efficacy of treatment regimens. The HCC827 cell line also supports a variety of dosing methods, including intraperitoneal, intravenous, and oral gavage administration, allowing for flexible and tailored therapeutic protocols. For more detailed investigation, additional techniques such as imaging and tumor site-specific injections can be employed. Comprehensive analyses, including tumor immunohistochemistry, molecular profiling, survival assessments, and histopathological evaluations, ensure a thorough assessment of therapeutic responses. The model also enables the evaluation of immune cell infiltration, angiogenesis, and changes in the tumor microenvironment. The HCC827 model is especially effective in studying the impact of EGFR-targeted therapies and exploring mechanisms of resistance, making it an essential tool in lung cancer research.
HCC827 Subcutaneous Lung Cancer Model
Subcutaneous HCC827 models are commonly used in preclinical studies to evaluate the efficacy of targeted therapies for non-small cell lung cancer (NSCLC). In this model, HCC827 cells, which harbor an EGFR exon 19 deletion mutation, are implanted subcutaneously into immunocompromised mice, typically in the flank area. The subcutaneous HCC827 model also provides valuable insights into the effectiveness of therapies targeting EGFR-driven tumors, given the cell line’s sensitivity to EGFR inhibitors like erlotinib. Tumor progression is monitored regularly using digital calipers, and treatment effects are evaluated based on tumor volume reduction, changes in body weight, and general health of the mice. This model can also be utilized to study resistance mechanisms that may emerge with long-term therapy, helping to guide the development of next-generation therapies. In addition to tumor size, post-mortem histological analysis is conducted to examine tumor architecture, cellular viability, and molecular markers associated with therapy response. The tumor microenvironment is also analyzed to assess immune cell infiltration and vascular changes induced by treatment.
NSCLC Metastasis (HCC827 Preclinical Model)
Metastatic HCC827 models are crucial for investigating the spread and progression of non-small cell lung cancer (NSCLC) driven by EGFR mutations, particularly exon 19 deletions. These models are typically established by injecting HCC827 cells intravenously, allowing the tumor cells to circulate and establish secondary growths in distant organs, such as the lungs, liver, or lymph nodes. Metastatic HCC827 models are valuable for understanding the mechanisms of tumor invasion and organ-specific metastasis in EGFR-mutant lung adenocarcinoma. They also serve as powerful tools for evaluating therapies targeting metastatic spread, including EGFR inhibitors, combination therapies, and potential novel agents. The models closely replicate the metastatic progression seen in advanced-stage NSCLC and provide insights into how tumors adapt to treatment. Advanced imaging techniques like bioluminescence and MRI are often employed to track metastatic lesions in real-time, offering a non-invasive approach for monitoring tumor burden. Histopathological analysis of metastatic sites enables the identification of cellular and molecular changes, shedding light on how therapies impact tumor progression. These models are particularly useful for preclinical studies focused on identifying novel treatment strategies and understanding the underlying biology of metastatic NSCLC.
Targeting EGFR in Lung Cancer with HCC827 Cells
The HCC827 cell line, derived from a lung adenocarcinoma, is characterized by its oncogene addiction to the epidermal growth factor receptor (EGFR). Specifically, it harbors a deletion mutation in exon 19 (Del E746-A750), a common sensitizing mutation associated with non-small cell lung cancer (NSCLC). This mutation leads to constitutive activation of the EGFR tyrosine kinase domain, driving uncontrolled proliferation and survival signaling. HCC827 cells are particularly sensitive to EGFR tyrosine kinase inhibitors (TKIs) like gefitinib, making them a widely used model for studying TKI efficacy and resistance mechanisms. The development of acquired resistance in this cell line is often linked to secondary mutations, such as T790M in exon 20, or alternative pathway activations, such as MET amplification. This cell line has been instrumental in exploring combination therapies and innovative strategies to overcome TKI resistance, including RNA-mediated and multitargeted approaches.
Oxymatrine Targets EGFR Signaling to Suppress NSCLC Growth in HCC827 Cells
A study conducted by Li W. et al., published in Cancer Medicine journal, examines the antitumor potential of oxymatrine, focusing on its effects on non-small cell lung cancer (NSCLC) and its suppression of the epidermal growth factor receptor (EGFR) signaling pathway. Oxymatrine was shown to inhibit EGFR activation and its downstream targets, including Akt and cyclin D1, leading to significant G0/G1 cell cycle arrest in HCC827 cells. In vivo experiments with HCC827 xenograft mouse models demonstrated that oxymatrine substantially suppressed tumor growth without observable toxicity. Mechanistically, oxymatrine’s inhibition of Akt signaling was critical, as exogenous Akt expression rescued cyclin D1 levels and reversed the cell cycle arrest. This study highlights HCC827 as a model for understanding EGFR-targeted therapies and underscores oxymatrine’s potential as a novel therapeutic agent for NSCLC.
Targeting HER3 to Enhance EGFR Inhibitor Efficacy in HCC827 NSCLC Models
Dr. Wu Y and colleagues published study in Molecular Cancer Therapeutics journal that explores the use of EZN-3920, a locked nucleic acid-based antisense oligonucleotide, to downregulate HER3 and enhance the effectiveness of EGFR and HER2 inhibitors in cancer treatment. EZN-3920 specifically inhibited HER3 mRNA and protein expression, resulting in reduced phosphorylation of HER3 and AKT, which are key drivers of tumor growth and survival. In xenograft models using HCC827 cells, a model for EGFR-overexpressing non-small cell lung cancer (NSCLC), EZN-3920 significantly suppressed tumor growth both alone and in combination with EGFR inhibitors like gefitinib. Notably, EZN-3920 also showed efficacy in gefitinib-resistant HCC827 cells, demonstrating its potential to overcome resistance mechanisms involving HER3. These findings underscore the importance of HER3 in NSCLC and highlight EZN-3920 as a promising therapeutic strategy for cancers reliant on HER3-mediated signaling pathways.
HCC827 Lung Cancer Subcutaneous and Metastatic Model: Download ![]()
