Lung cancer is one of the leading causes of cancer mortality. Lung cancer is primarily classified into two major histological subtypes: small cell lung cancer (SCLC) and non-small cell lung cancer (NSCLC), with NSCLC comprising approximately 85% of cases. The NSCLC is further subdivided into adenocarcinoma, squamous cell carcinoma, and large cell carcinoma, each exhibiting distinct molecular and clinical features that influence treatment strategies. Xenograft models are powerful research tool that involve the implantation of human tumor tissue or cells into immunodeficient animal models and serve as valuable tools for studying cancer biology and evaluating therapeutic interventions. By closely monitoring tumor growth and response to therapy in xenograft models, researchers can identify promising therapeutic candidates and better predict clinical outcomes. Xenografts play a critical role in preclinical cancer research, providing insights that bridge laboratory-based studies and clinical trial development. The NCI-H1993 (H1993) cell line is an epithelial-derived culture established from a 47-year-old white female patient diagnosed with stage 3A non-small cell lung cancer (NSCLC), specifically adenocarcinoma. This cell line is characterized by its epithelial morphology and provides a valuable in vitro model for studying the molecular mechanisms underlying NSCLC progression. As a representation of advanced-stage lung cancer, H1993 cells exhibit key genetic and phenotypic features that make them a useful tool for drug screening, therapeutic research, and the exploration of resistance mechanisms. The cell line has also been employed in studies focused on identifying novel molecular targets for treatment and evaluating the efficacy of potential anticancer compounds. Additionally, the use of H1993 cells can contribute to investigations into the tumor microenvironment and the interactions between cancer cells and stromal components. The H1993 xenograft model provides a robust platform for evaluating therapeutic agents targeting non-small cell lung cancer (NSCLC) with MET gene amplification in preclinical studies. At Altogen Labs, key experimental endpoints include Tumor Growth Delay (TGD) that offer critical insights into the efficacy of investigational therapies. The H1993 model supports flexible dosing regimens, including intravenous, intraperitoneal, intratumoral, and oral gavage administration, allowing for the design of tailored treatment protocols. For metastasis studies, alternative methods such as orthotopic implantation or tail vein injections can be employed to mimic advanced disease stages. Comprehensive evaluations include tumor immunohistochemistry, survival analysis, gross necropsy, and detailed histopathology, enabling a thorough assessment of therapeutic responses. The H1993 model is particularly effective for investigating the efficacy of MET-targeted therapies and combination treatments, as well as exploring the molecular mechanisms underlying tumor growth and resistance in NSCLC.
H1993 Orthotopic and Metastatic Model: Download ![]()
Studying Lung Cancer using Orthotopic H1993 Xenograft Model
Orthotopic H1993 models involve the implantation of H1993 cells directly into the lungs of immunocompromised mice, creating a biologically relevant microenvironment to study non-small cell lung cancer (NSCLC). These models more accurately recapitulate the tumor’s growth, interactions with the lung microenvironment, and potential for spontaneous metastasis compared to subcutaneous models. Orthotopic models are ideal for evaluating MET-targeted therapies, as the H1993 cell line is highly dependent on MET signaling for proliferation and survival. Tumor progression can be monitored using advanced imaging techniques, enabling longitudinal studies of therapeutic efficacy. Histological and molecular analyses of primary lung tumors and metastatic lesions provide insights into treatment-induced effects on tumor architecture, angiogenesis, and signaling pathways. These models are particularly valuable for studying drug resistance and testing novel combination therapies in a setting that closely resembles human lung cancer biology. The orthotopic H1993 model serves as a powerful tool for preclinical research, bridging the gap between in vitro studies and clinical applications.
Key Mutations and Oncogenic Traits in H1993 NSCLC Cells
H1993 is a human lung cancer cell line that exhibits oncogenic characteristics commonly observed in non-small cell lung cancer (NSCLC). It is known to harbor mutations in key genes such as KRAS and TP53, which are critical drivers of tumorigenesis. The cells demonstrate enhanced proliferation, resistance to apoptosis, and the ability to invade surrounding tissues, all of which contribute to their aggressive behavior. Additionally, MET gene dysregulation, through overexpression or mutations, plays a significant role in driving cell proliferation, migration, and invasiveness, further supporting the tumor’s growth and metastatic potential. H1993 cells also exhibit increased angiogenesis, providing the tumor with the necessary blood supply to support rapid growth. They undergo epithelial-to-mesenchymal transition (EMT), a process associated with metastasis, further enhancing their ability to spread to distant sites. These characteristics make H1993 a useful model for studying lung cancer biology and testing potential therapeutic strategies.
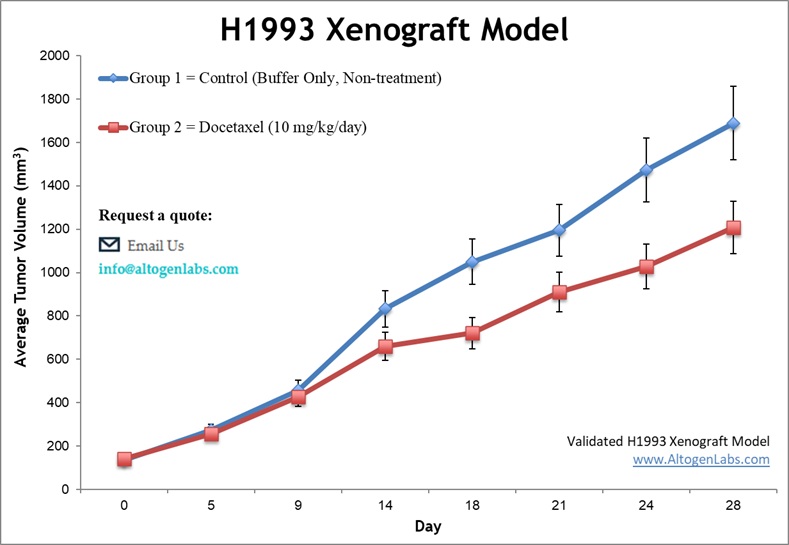
H1993 Subcutaneous Xenograft Model for Preclinical Research
The subcutaneous H1993 xenograft model involves the implantation of H1993 tumor cells, derived from human lung adenocarcinoma with MET gene amplification, into immunocompromised mice to study non-small cell lung cancer (NSCLC). These models are established by injecting the tumor cells subcutaneously into the immunocompromised mice, allowing for reproducible monitoring of tumor growth and therapeutic responses. This model provides a robust platform for evaluating the efficacy of MET-targeted therapies and investigating the molecular mechanisms underlying tumor progression and drug resistance. The subcutaneous location enables straightforward, non-invasive measurement of tumor volume, making it suitable for longitudinal studies on treatment efficacy. Frequently utilized in preclinical research, the NCI-H1993 xenograft model supports the development of novel therapies while complementing more complex orthotopic and metastatic models for translational cancer studies.
Antitumor Mechanisms of Gambogic Acid in MET-Amplified NSCLC
A study done by Li D, et al., published by the Oncology Letters journal discussed the antitumor mechanisms of gambogic acid (GA) using NCI-H1993 xenograft models of non-small cell lung cancer (NSCLC), which harbor a MET gene amplification. The researchers administered varying doses of GA to mice and observed a dose-dependent inhibition of tumor growth without significant toxicity. GA was found to downregulate the expression of phosphorylated MET (p-MET) and its downstream signaling molecules, p-AKT and p-ERK, as shown through western blot analysis. Immunohistochemical staining revealed reduced Ki-67 expression, indicating inhibited tumor cell proliferation. From this study, researchers suggest GA’s potential as a therapeutic agent targeting MET-amplified NSCLC, emphasizing its ability to modulate the MET signaling pathway effectively.
Metastatic H1993 Models: Advancing NSCLC Research and Therapy
Metastatic H1993 models are valuable tools for studying the dissemination of non-small cell lung cancer (NSCLC) with MET amplification, providing insights into the mechanisms of tumor invasion and organ-specific metastasis. These models are typically established by injecting H1993 cells intravenously or orthotopically, allowing tumor cells to circulate and colonize distant organs such as the lungs, liver, or bones. Metastatic models closely mimic the progression of advanced-stage lung cancer and are instrumental in evaluating therapies that target metastatic spread, including anti-MET agents, immunotherapies, and anti-angiogenic drugs. Researchers often monitor metastatic lesions through advanced imaging techniques, such as bioluminescence or fluorescence imaging, to track tumor burden in real-time. Histological analyses of metastatic sites provide additional insights into cellular and molecular changes induced by treatment. These models are particularly useful for assessing combination therapies and studying biomarkers associated with metastatic progression, offering translational relevance for clinical applications.
H1993 Orthotopic and Metastatic Model: Download ![]()
