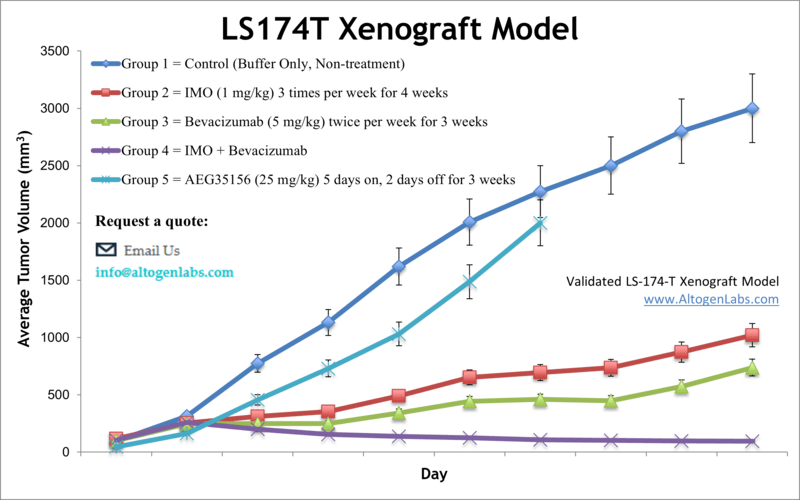
LS174T xenograft model
Colorectal carcinoma remains one of the most prevalent malignancies in the United States, and xenograft models derived from human colorectal tumors are essential for preclinical evaluation of novel therapeutic agents. The LS174T cell line, established from a Duke’s type B colorectal adenocarcinoma of a 58-year-old female patient, is widely used in colon cancer research due to its well-characterized molecular and morphological features. LS174T cells express multiple oncogenes, including c-myc, N-myc, H-ras, N-ras, Myb, and fos, and display abundant microvilli, cytoplasmic vacuoles, and prominent mucin secretion. This cell line produces high levels of carcinoembryonic antigen (CEA), interleukin-6 (IL-6), interleukin-10 (IL-10), and mucin, which contribute to its relevance in studies involving tumor-associated glycoproteins and immunomodulatory cytokines. The LS174T xenograft model has been instrumental in evaluating mucin-targeted cancer therapies, as demonstrated by early studies characterizing tumor-specific glycosylation patterns of secreted mucins that may serve as drug delivery targets. Additionally, LS174T tumors have been used to assess angiogenesis inhibitors such as YN968D1, a potent VEGFR2, Ret, and c-Kit tyrosine kinase inhibitor with strong antitumor activity. In vivo imaging studies utilizing LS174T cells have validated the application of radiolabeled antibodies against tumor-associated glycoprotein-72 (TAG-72), highlighting the potential for PET-guided diagnostic and therapeutic strategies. Further investigations have shown that rapamycin resistance in LS174T tumors is mediated through JNK pathway activation, and that concurrent JNK inhibition enhances the antiproliferative efficacy of mTOR-targeted agents. LS174T cells are commonly used to generate subcutaneous CDX models in immunodeficient mice and are highly suitable for studying both monotherapy approaches, including RNAi-based interventions, and combination regimens designed to overcome resistance to agents such as bevacizumab through immunostimulatory pathways like TLR9 activation. A 2011 study used LS174 colon cancer cells to investigate the contribution of c-Jun N-terminal kinase (JNK) signaling to rapamycin resistance.
LS174 Colorectal Cancer Xenograft Model: Download ![]()
Download Altogen Labs LS174T Xenograft Model PowerPoint Presentation: ![]()
LS174T Cell Line
The LS174T cell line, derived from a human colorectal adenocarcinoma, serves as a widely utilized in vitro and in vivo model for investigating mucin production, intestinal epithelial biology, and colorectal cancer pathogenesis. A defining feature of LS174T cells is their constitutive expression of MUC2, a secretory mucin that is critical to the intestinal mucosal barrier, rendering the line particularly relevant for studies of barrier function in mucinous tumors. These cells harbor activating mutations in KRAS (G12D) and inactivating alterations in TP53, both of which contribute to tumor proliferation, survival, and resistance to standard chemotherapeutic agents such as 5-fluorouracil and irinotecan. LS174T cells also exhibit a partially differentiated epithelial phenotype and heterogeneous expression of stemness-associated markers including CD44 and LGR5. The cell line displays dysregulated Wnt signaling due to APC mutation, resulting in elevated transcription of downstream oncogenes such as MYC and CCND1. In xenograft models, LS174T tumors demonstrate moderate growth kinetics and maintain their mucin-secreting properties in vivo.
Subcutaneous LS174T Xenografts in Colorectal Cancer Research
Subcutaneous xenograft transplantation using LS174T cells is a well-established method for modeling human colorectal cancer in vivo, offering a reproducible and accessible platform for investigating tumor growth dynamics, molecular signaling, and therapeutic response. LS174T, a mucin-producing colorectal adenocarcinoma cell line harboring KRAS and TP53 mutations, reliably forms moderately growing, mucin-rich tumors when injected subcutaneously into immunocompromised mice such as NOD/SCID or athymic nude strains. These tumors preserve essential histological features of mucinous colorectal carcinoma, making the model particularly suitable for evaluating agents that target epithelial differentiation, Wnt signaling, or mucin-associated barriers. Although subcutaneous models lack the anatomical and stromal complexity of the native colonic environment, their technical simplicity facilitates high-throughput screening, longitudinal tumor monitoring, and efficient collection of tissue for molecular and histopathological analysis.
Ongoing research using LS174T subcutaneous xenografts increasingly explores the relationship between mucin secretion, immune exclusion, and drug resistance. The dense extracellular matrix and high MUC2 expression characteristic of this model are believed to limit both therapeutic penetration and immune cell infiltration, underscoring the relevance of combinatorial strategies involving mucolytic agents or immune modulators. While the absence of a fully functional immune microenvironment remains a limitation, emerging approaches such as co-engraftment with stromal or immune components offer promising avenues to enhance physiological relevance. The LS174T xenograft model therefore represents a valuable experimental system for advancing the understanding of mucinous colorectal cancer and for evaluating the efficacy of novel therapeutic approaches.
Antibody Penetration Barriers in LS174T Xenografts
In a study by Coutelle O, et al., published in the British Journal of Cancer, the authors evaluate the tumor response of LS174T colorectal cancer xenografts to radioimmunotherapy and examine the spatial limitations of antibody distribution within structurally complex tumor environments. LS174T, a human colorectal adenocarcinoma cell line characterized by mucinous histology and oncogenic mutations in KRAS and TP53, demonstrates restricted therapeutic efficacy due to non-uniform antibody penetration. Although radiolabeled anti-CEA antibody (A5B7) was successfully delivered systemically, intratumoral accumulation remained confined to perivascular regions, leaving substantial portions of tumor tissue untreated. This pattern of localization reflects a significant barrier posed by the tumor microarchitecture, which includes irregular vasculature and patchy CEA expression.
The authors identify a strong correlation between vascular disorganization and the binding site barrier, where high-affinity antibodies saturate proximal antigen sites, preventing diffusion into distal tumor zones. In comparison to SW1222 xenografts, which exhibit organized glandular architecture and uniform CEA expression, LS174T tumors displayed limited antibody distribution and reduced therapeutic benefit. High-resolution imaging methods, including phosphor autoradiography, immunofluorescence, and transmission electron microscopy, effectively mapped the spatial interactions between vasculature, antigen, and antibody. Despite the study’s strength in visualizing microanatomical constraints, it is limited by its focus on two xenograft models and use of immunodeficient mice, excluding potential immune-mediated effects. The findings underscore the importance of tumor microenvironment and structural barriers in determining therapeutic response. To improve outcomes in mucinous colorectal cancers such as those modeled by LS174T, future studies should investigate strategies that enhance antibody diffusion, modulate stromal composition, or incorporate immune-competent and orthotopic systems to capture a more complete therapeutic profile.
Microenvironmental Constraints on LS174T Radioimmunotherapy
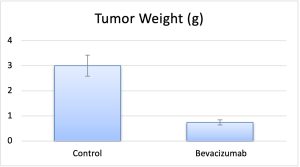
The LS174T colorectal cancer xenograft model displays a moderate to poorly differentiated histology with disorganized tumor architecture and a highly heterogeneous vascular network. Compared to the more glandular and vascularized SW1222 model, LS174T tumors demonstrated markedly uneven distribution of blood vessels and carcinoembryonic antigen (CEA), with antigen primarily localized to perivascular tumor cells. Despite similar overall antibody uptake, LS174T required a threefold higher dose of radiolabeled anti-CEA antibody (A5B7) to achieve equivalent tumor growth inhibition, highlighting the critical influence of microanatomical features on therapeutic efficacy. Fluorescence and phosphor plate imaging confirmed that in LS174T tumors, antibodies penetrated minimally beyond the perivascular space and remained sequestered near the vasculature, resulting in large untreated regions. This limited dispersion was attributed not to interstitial pressure or gap junctions, but to the so-called binding site barrier; an effect whereby high-affinity antibodies bind rapidly to nearby antigens, preventing broader diffusion. In contrast, SW1222 tumors supported more uniform antibody movement and retention, aided by expansive intercellular spaces and consistent antigen accessibility.
Chemotherapy Efficacy in Mucinous LS174T Tumors
The LS174T human colorectal adenocarcinoma cell line, when established as subcutaneous xenografts in immunocompromised mice, exhibits differential sensitivity to standard chemotherapeutic agents. Among the treatments evaluated, irinotecan produced the most sustained tumor suppression, with clear dose-dependent effects and strong associations with increased apoptosis and reduced cellular proliferation. In contrast, 5-fluorouracil and oxaliplatin induced only modest and transient reductions in tumor growth, highlighting the selective efficacy of irinotecan in this model. Histological analysis further supported these findings, revealing significantly greater tumor necrosis and caspase-3 activation in the irinotecan-treated group. These patterns suggest that LS174T xenografts are particularly responsive to topoisomerase I inhibition, although incomplete tumor regression and residual viable tumor areas point to underlying resistance mechanisms. The structural features of LS174T tumors, including a dense extracellular matrix and mucinous architecture, may hinder drug penetration and contribute to spatial heterogeneity in treatment response. This microenvironmental barrier is likely a key factor in the persistence of resistant cell populations despite systemic chemotherapy. The study’s design, which included longitudinal tumor measurements and endpoint analyses such as immunohistochemistry and semi-quantitative response scoring, provided a robust framework for assessing drug efficacy. However, limitations such as small sample sizes and the use of immunodeficient mouse hosts restrict the generalizability of the results. To build on these findings, future research should explore combination therapies that improve drug delivery or counteract resistance, and incorporate orthotopic or immune-competent models to better replicate the native tumor environment. The LS174T xenograft remains a relevant model for advancing the preclinical evaluation of therapeutic strategies targeting mucinous colorectal cancer.
Oncogenic Signatures in LS174T Colorectal Cancer Cells
The LS174T human colorectal adenocarcinoma cell line presents a distinctive oncogenic profile, marked by mutations and dysregulation in several key pathways implicated in colorectal tumorigenesis. Prominently, LS174T cells harbor a KRAS mutation at codon 12 (G12D), which constitutively activates the MAPK signaling cascade, promoting unchecked cellular proliferation and survival. Additionally, TP53 is inactivated, compromising DNA damage responses and facilitating genomic instability. The expression of MYC, a downstream effector of both Wnt and MAPK signaling, is upregulated and plays a critical role in driving cell cycle progression and metabolic reprogramming. Notably, LS174T cells also demonstrate elevated levels of MUC2, a secretory mucin characteristic of mucinous colorectal carcinomas, which contributes to the formation of a dense extracellular matrix and impacts drug delivery and immune cell infiltration. An important observation is the preserved activation of the canonical Wnt/β-catenin pathway in LS174T cells due to mutations in APC, a key negative regulator of β-catenin stability. This leads to increased nuclear localization of β-catenin and subsequent transcription of target genes such as CCND1 and LGR5, which are involved in stemness and cell cycle control. The interplay between KRAS-driven MAPK activation and Wnt pathway dysregulation reflects a convergence of signaling pathways that together support tumor progression, resistance to therapy, and epithelial plasticity. The methodology used to identify these signatures involved RNA sequencing, quantitative PCR, and Western blot analysis, providing multi-level validation of gene expression. While robust in design, the findings are limited by the absence of in vivo confirmation and the reliance on a single cell line model. Still, the molecular landscape of LS174T offers a valuable framework for investigating therapeutic targets in mucinous and KRAS-mutant colorectal cancers. Future research should prioritize functional studies using gene knockdown or CRISPR-based editing, as well as the development of co-culture and xenograft models to elucidate how these oncogenic drivers influence tumor behavior in complex biological environments.
Patient-Derived Organoids in Precision Cancer Research
Organoids are three-dimensional in vitro culture systems derived from patient tumor samples that faithfully preserve the genetic, phenotypic, and architectural complexity of the original malignancy. Unlike conventional two-dimensional cell cultures, organoids retain multicellular organization, differentiation gradients, and clonal heterogeneity, making them highly relevant for modeling tumor biology. They can be expanded long-term from primary tissue, enabling scalable experimentation and individualized therapeutic profiling. While in vivo models such as xenografts and allografts remain essential for capturing stromal and immune interactions, organoids offer a rapid and high-throughput alternative for assessing drug response and resistance mechanisms. The development of patient-derived tumor organoid (PDTO) biobanks has further advanced the field, providing a living resource for investigating tumor heterogeneity, identifying molecular vulnerabilities, and accelerating the discovery of precision oncology therapies.
LS174 Colorectal Cancer Xenograft Model: Download ![]()
Basic study design
- Cells are collected and viability is determined. Trypan blue is utilized and a 98-99% minimum viability is required for study initiation.
- Each mouse (NOD/SCID or BALB/C athymic, 10-12 weeks old) receive a single s.c. injection. One million cells is injected into the flank of a hind leg (volume of 100 µL) of the Matrigel cell suspension.
- Tumors are then measured with digital calipers. An average size of 50-150 mm3are needed to begin the study. Animals are then sorted into groupings that best represent randomization. Compounds are administered following the treatment program.
- On-study measurements include tumor volume and mouse weights. A necropsy is performed at the end of the in-life portion to remove tumors, record tumor weight and document digitally.
- Tissues are then submersed in RNA-later reagent, or nucleic acids isolated, tumor snap frozen or prepared for histology.
Get Instant Quote for
LS174T Xenograft Model
Xenograft animal models are used to assess the effectiveness of drugs against specific types of cancer. New medicines are tested on staged tumor growths that have been engrafted via subcutaneous or orthotopic inoculation in an immunocompromised mouse or rat model. All clinically approved anti-cancer agents have been evaluated with conventional preclinical in vivo models. Xenograft studies can be highly complex, starting with the selection of the appropriate animal model, choice of tumorigenic cell line, administration method, dosing, analysis of tumor growth rates and tumor analysis (histology, mRNA and protein expression levels). The dosing of the experimental compound of interest is initiated, for a staged study, when the mean tumor size reaches a specified volume (typically 75-120 mm3). In an unstaged study, the dosing of the compound of interest is initiated immediately after xenografting.
Animal handling and maintenance at the Altogen Labs facility is IACUC-regulated and GLP-compliant. Following acclimatization to the vivarium environment, mice are sorted according to body mass. The animals are examined daily for tumor appearance and clinical signs. We provide detailed experimental procedures, health reports and data (all-inclusive report is provided to the client that includes methods, results, discussion and raw data along with statistical analysis). Additional services available include collection of tissue, histology, isolation of total protein or RNA and analysis of gene expression. Our animal facilities have the flexibility to use specialized food or water systems for inducible gene expression systems.
Following options are available for the LS174T xenograft model:
- LS174T Tumor Growth Delay (TGD; latency)
- LS174T Tumor Growth Inhibition (TGI)
- Dosing frequency and duration of dose administration
- Dosing route (intravenous, intraperitoneal, intratumoral, oral gavage, intramuscular, subcutaneous)
- LS174T tumor immunohistochemistry
- Blood chemistry analysis
- Toxicity and survival (optional: performing a broad health observation program)
- Gross necropsies and histopathology
- Positive control group employing dox or cyclophosphamide, at typical dosages of 20-40 mg/kg can be administered i.v. or by intramuscular injection to the control group QOD for the study duration
- Lipid distribution and metabolic assays
