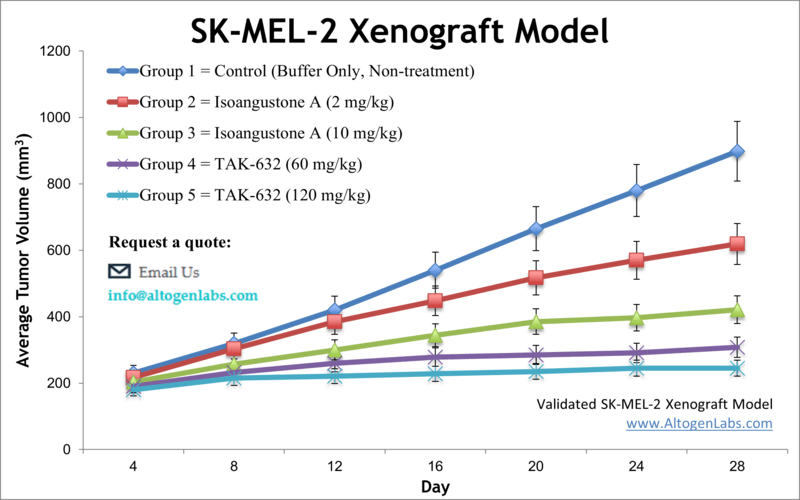
SK-MEL-2 xenograft model
The SK-MEL-2 cell line is a commonly used human melanoma cancer cell line that was originally derived from a tumor in the skin of a 54-year-old male patient. The SK-MEL-2 cell line is known to have mutations in the BRAF and NRAS genes, which are common genetic alterations found in melanoma. Human malignant melanoma is a highly metastatic and drug-resistant form of cancer that originates from the transformation of melanocytes. Despite recent advances in melanoma research, new treatment approaches targeting NRAS-mutated melanoma are required. The SK-MEL-2 tumorigenic cell line was established in 1972 from the skin of a 60-year-old Caucasian male patient with malignant melanoma. SK-MEL-2 expresses mutant N-Ras and wildtype B-Raf. A 2014 study by Wada et al. published in PLoS One investigated the activity of a potent and selective dual RAF/MEK inhibitor CH5126766/RO5126766 using the SK-MEL-2 xenograft model. The article reports that CH5126766/RO5126766 blocks the tumor growth in the SK-MEL-2 xenograft model and is an attractive RAF/MEK inhibitor in RAS-mutated cancer cells including melanoma. Blackham et al. released a Surgery article in 2013 characterizing the mechanism of susceptibility of SK-Mel xenograft melanoma cells to mutant M protein vesicular stomatitis virus (M51R-VSV). Results demonstrated that M51R-VSV decreased tumor growth in xenograft models but with significant variance. A 2013 PNAS article (Posch et al.) used the SK-MEL-2 model to study the influence of MAP, ERK, MEK, PI3K and mTOR signaling cascades in neuroblastoma rat sarcoma viral oncogene homolog (NRAS) mutant melanoma cells, which are notorious for lack of effective therapeutic treatment. Results demonstrated that combination targeting of MEK and PI3K/mTOR pathways is effective in causing xenograft tumor regression and inhibiting in vitro growth. Finally, Claffey et al. published a study in Cancer Research (1996) using the SK-Mel-2 xenograft model to characterize the role of vascular permeability factor (VPF) and VEGF in tumors, which are now a common therapeutic target. Results demonstrated that VPF/VEGF expression promotes tumor angiogenesis, growth and metastasis. The SK-MEL-2 cell line (human melanoma) is used to create the CDX (Cell Line Derived Xenograft) SK-MEL-2 xenograft mouse model. The SK-MEL-2 xenograft model is an established preclinical model enabling studies on anti-tumor activity of MEK, PI3K and mTOR inhibitors in both mono and combination therapies.
Download Altogen Labs SKMEL2 Xenograft Model PowerPoint Presentation: ![]()
Basic study design
- Initially, cells are collected via trypsin-EDTA and viability is determined. One million cells (SK-MEL-2 plus 50% Matrigel) in an injection volume of 100-120 µL is administered into 11-12 week old athymic BALB/C Nude mice subcutaneously.
- The test article administration begins as tumor sizes are in the range of ~100 mm3.
- Study groups are established and the treatment cohorts are injected with test material following the dose schedule. Pre- and post-dose tumor size measurements are recorded, including whole body weights and any associated body weight loss (BWL).
- After final dose, mice are euthanized and tissues excised for further analysis. Tumors are removed and weighed. All tissues of interest can be immersed in RNAlater reagent, snap frozen or placed in 10% NBF formalin.
Get Instant Quote for
SK-MEL-2 Xenograft Model
Xenograft animal models are used to assess the effectiveness of drugs against specific types of cancer. New medicines are tested on staged tumor growths that have been engrafted via subcutaneous or orthotopic inoculation in an immunocompromised mouse or rat model. All clinically approved anti-cancer agents have been evaluated with conventional preclinical in vivo models. Xenograft studies can be highly complex, starting with the selection of the appropriate animal model, choice of tumorigenic cell line, administration method, dosing, analysis of tumor growth rates and tumor analysis (histology, mRNA and protein expression levels).
The dosing of the experimental compound of interest is initiated, for a staged study, when the mean tumor size reaches a specified volume (typically ~100 mm3). Mice are dosed once or twice a day for 28 days (or other desired study duration) via the chosen route of administration. Tumor volume (mm3) is calculated via the “(W x W x L) / 2” formula, where W is tumor width and L is tumor length.
Following options are available for the SK-MEL-2 xenograft model:
- SK-MEL-2 Tumor Growth Delay (TGD; latency)
- SK-MEL-2 Tumor Growth Inhibition (TGI)
- Dosing frequency and duration of dose administration
- Dosing route (intravenous, intratracheal, continuous infusion, intraperitoneal, intratumoral, oral gavage, topical, intramuscular, subcutaneous, intranasal, using cutting-edge micro-injection techniques and pump-controlled IV injection)
- SK-MEL-2 tumor immunohistochemistry
- Alternative cell engraftment sites (orthotopic transplantation)
- Blood chemistry analysis
- Gross necropsies and histopathology
- Positive control group employing cyclophosphamide, at a dosage of 30-40 mg/kg
