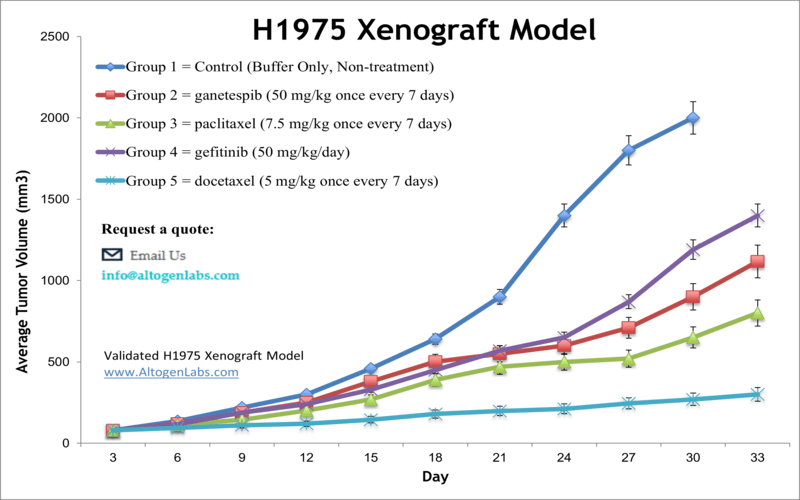
NCI-H1975 xenograft model
The NCI-H1975 epithelial cell line was isolated in 1988 from lung cells taken from a non-smoker female patient with adenocarcinoma. This cell line is a potent tool for performing preclinical tests for lung cancer treatment. Although lung cancer therapeutics have evolved dramatically within last two decades, lung cancer is considered the primary cause of cancer deaths globally. Non-small cell lung cancer (NSCLC) is responsible for the majority of lung cancer cases. New approaches in lung cancer therapy are vital in treating NSCLC patients. A study by Smith et al. published in Targeted Oncology investigated the antitumor efficacy of selective EGFR tyrosine kinase inhibitors (TKIs) and the HSP90 inhibitor ganetespib, alone and in combination, using the NCI-H1975 xenograft model. The results indicate that concurrent administration of ganetespib overcomes erlotinib resistance and significantly improves tumor growth inhibition in erlotinib-resistant NCI-H1975 xenografts. According to the article, combination treatment with both drugs substantially enhances antitumor response and could be a therapy of choice for NSCLC patients. A 2018 Clinical Cancer Research study (Steiner et al.) used the H1975 xenograft model to study the effects of mutated epidermal growth factor receptor (EGFR) with treatment with cetuximab, a monoclonal IgG1 antibody also known as Erbitux. EGFR is mutated (somatic) in 10% of patients which affects EGFR targeted therapy; results demonstrated that cetuximab had antitumor activity with both wild type and mutated EGFR and that combination treatments with cisplatin or docetaxel increased these effects which is particularly relevant for chemorefractory NSCLC. In 2015 Cross et al. released a study in Cancer Discovery using the NCI-H1975 model to look at overcoming T790-mediated resistance to EGF receptor tyrosine kinase inhibitors (EGFR TKIs). They found that treatment with AZD9291, an irreversible dual inhibitor of EGFRm+ and T790M inhibits tumor cell growth and signaling pathways in advanced NSCLC. The H1975 cell line (human lung) is used to create the CDX (Cell Line Derived Xenograft) NCI-H1975 xenograft mouse model. The H1975 xenograft model is a mutated EGFR (T790M, L858R) expressing model used for preclinical studies of monotherapies or in combination (e.g. cetuximab, cisplatin, gemcitabine, docetaxel).
Download Altogen Labs NCI-H1975 Xenograft Model PowerPoint Presentation: ![]()
Basic study design
- Prior to trypsinization of H1975 cells, they are continually grown at a phase of exponential growth. Viable cell counts are determined using trypan blue and concentration is adjusted to appropriate density needed for xenotransplantation.
- 11-12 week old athymic BALB/C mice receive subcutaneous injection into the hind leg. Each animal receives a 160 µL injection containing one million cells of the Matrigel + H1975 cell suspension.
- Tumors are calipered until growth reaches average sizes of 75-150 mm3. At this point, grouping of mice into treatment cohorts and injections of the compound of interest is started according to treatment schedule.
- Mouse body weights are documented up to 2-3 times weekly, with tumors calipered daily.
- When the end of study parameters are reached (i.e. maximum tumor size), animals are euthanized. All tissues collected snap frozen, submerged in a stabilizing solution (e.g. RNA Later reagent) or nucleic acids isolated. Tumors are excised from the mice and weighed.
Get Instant Quote for
NCI-H1975 Xenograft Model
All clinically approved anti-cancer agents have been evaluated with conventional preclinical in vivo models. Xenograft studies can be highly complex, starting with the selection of the appropriate animal model, choice of tumorigenic cell line, administration method, dosing, analysis of tumor growth rates and tumor analysis (histology, mRNA and protein expression levels).Xenograft animal models are used to assess the effectiveness of drugs against specific types of cancer. New medicines are tested on staged tumor growths that have been engrafted via subcutaneous or orthotopic inoculation in an immunocompromised mouse or rat model. Altogen Labs provides an array of laboratory services using over 120 validated CDX and PDX models. Researchers investigating the role of specific proteins or gene products in regulating tumor growth can benefit from development of protein overexpression (genetically engineered to ectopically express proteins, tumor suppressors, or oncogenes) and RNAi cell lines with long term gene silencing. Altogen Labs provides quantitative gene expression analysis of mRNA expression (qPCR) and protein expression analysis using the WES system (ProteinSimple). We provide detailed experimental procedures, health reports and data (all-inclusive report is provided to the client that includes methods, results, discussion and raw data along with statistical analysis). Additional services available include collection of tissue, histology, isolation of total protein or RNA and analysis of gene expression.
Following options are available for the H1975 xenograft model:
- H1975 Tumor Growth Delay (TGD; latency)
- H1975 Tumor Growth Inhibition (TGI)
- Dosing frequency and duration of dose administration
- Dosing route (intravenous, intratracheal, continuous infusion, intraperitoneal, intratumoral, oral gavage, topical, intramuscular, subcutaneous, intranasal, using cutting-edge micro-injection techniques and pump-controlled IV injection)
- H1975 tumor immunohistochemistry
- Alternative cell engraftment sites (orthotopic transplantation, tail vein injection and left ventricular injection for metastasis studies, injection into the mammary fat pad, intraperitoneal injection)
- Blood chemistry analysis
- Toxicity and survival (optional: performing a broad health observation program)
- Gross necropsies and histopathology
- Positive control group employing cyclophosphamide, at a dosage of 50 mg/kg administered by intramuscular injection to the control group daily for the study duration
- Lipid distribution and metabolic assays
- Imaging studies: Fluorescence-based whole body imaging
