MKN45 xenograft model
MKN45 cells are a human gastric cancer cell line that was established from the biopsy specimen of a patient with gastric adenocarcinoma. These cells are widely used as an in vitro model of gastric cancer to study the mechanisms underlying tumor development and to test the efficacy of potential therapeutic agents. MKN45 cells exhibit many of the characteristics of gastric cancer cells, including the ability to form colonies in vitro and to form tumors when injected in vivo into immunocompromised mice. These cells have been extensively studied to better understand the molecular mechanisms underlying gastric cancer development and progression. MKN-45 cells have been used to study the role of various signaling pathways in gastric cancer, including the Wnt/β-catenin, PI3K/Akt, and MAPK/ERK pathways. These pathways are known to be dysregulated in many types of cancer, including gastric cancer, and targeting them with therapeutic agents has shown promise in preclinical studies. MKN-45 cells have also been used to test the efficacy of various chemotherapeutic agents, including cisplatin, 5-fluorouracil, and docetaxel. In addition, these cells have been used to develop and test novel targeted therapies, such as monoclonal antibodies and small molecule inhibitors.
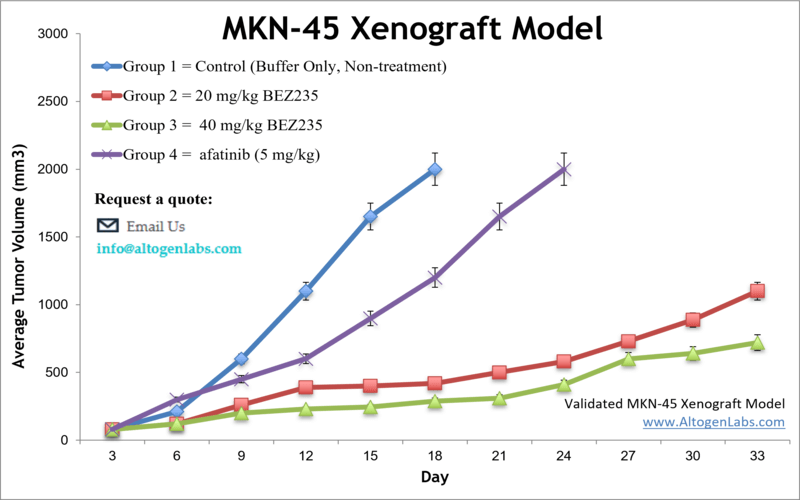
Gastric cancer is still a common cause of cancer-related deaths worldwide and often diagnosed at an advanced stage. Per the American Cancer Society (ACS) approximately 1 in 111 people may develop gastric cancer, the third primary cause of cancer mortality in the world. The MKN-45 cell line was established in Japan from a poorly differentiated adenocarcinoma of a 62-year-old woman. The MKN-45 cell line expresses wild-type p53 protein and is characterized by amplification of c-met oncogene and promoter mutation of E-cadherin. Preclinical murine models of gastric cancer help researchers to evaluate management strategies for stomach cancer. A 2011 study published in Clinical Cancer Research, tested the antitumor activity of the PI3K/mTOR kinase inhibitor BEZ235 against gastric cancer both in vitro and in vivo. The article indicates that the MKN-45 xenograft model is resistant to treatment with BEZ235 in vivo. The 2013 study by Lio et al. characterized the MKN-45 cell line cultured in enriched medium and identified spheroid body-forming cells as well as overexpression of cancer stem cell (CSC) markers (CD44, Nanog, Oct4, Sox2). Identifying CSCs, which possess properties that contribute to tumor formation, metastasis and recurrence, in gastric cancer models is considered important for developing stem cell-targeting therapies. A 2012 study by Yamashita-Kashima et al. (2012) used MKN-45 cells to look for biomarkers indicative of anticancer activity of bevacizumab, a monoclonal antibody against human endothelial cell growth factor (VEGF) used for chemotherapy. The group concluded that VEGF levels and VEGF/bFGF ratios were most correlated to bevacizumab sensitivity but that other predictive markers are needed for this model. The last example study using MKN45 cells is the 2010 study by Chong et al. which screened for biomarkers of gastric tumor growth and found that in the MKN45 xenograft model reduced plasma levels of human APOA1 correlated to tumor size. This has potential clinical relevance in terms of tumor detection. The MKN-45 cell line is used to create the CDX model of gastric cancer. The MKN-45 xenograft model is also resistant to TGF-B growth inhibition, carboplatin, docetaxel and irinotecan. However, there is anti-tumor activity as evident from afatinib and BEX235.
Download Altogen Labs MKN-45 Xenograft Model PowerPoint Presentation: ![]()
Basic study design
- Exponentially growing MKN-45 cells are collected and cell viability is determined utilizing trypan blue. The concentration of the cell suspension is adjusted to the appropriate density.
- Athymic BALB/C or NOD/SCID mice (10 to 12 w.o.) receive single, subcutaneous injection of one million cells (injection volume of 100-150 microliters with matrigel).
- Tumor growth is measured by calipering until an average size of 80-120 mm3 reached. Animals are then randomized to form the treatment cohorts. The test compound is administered according to the client supplied treatment schedule. Tumor sizes are measured daily.
- Animals are euthanized as tumor size reaches 2,000 cu millimeters. A final necropsy and tissue collection is performed. Tumors are excised from the animals and weighed. Optional digital imaging of tumors is available.
- Standard gross necropsies are completed; tissues are collected for downstream analysis.
Get Instant Quote for
MKN-45 Xenograft Model
Xenograft animal models are used to assess the effectiveness of drugs against specific types of cancer. New medicines are tested on staged tumor growths that have been engrafted via subcutaneous or orthotopic inoculation in an immunocompromised mouse or rat model. All clinically approved anti-cancer agents have been evaluated with conventional nonclinical in vivo models. Xenograft studies can be highly complex, starting with the selection of the appropriate animal model, choice of tumorigenic cell line, administration method, dosing, analysis of tumor growth rates and tumor analysis (histology, mRNA and protein expression levels).
The dosing of the experimental compound of interest is initiated, for a staged study, when the mean tumor size reaches a specified volume (typically 80-120 mm3). In an unstaged study, the dosing of the compound of interest is initiated 3-5 days post xenografting. Animal handling and maintenance at the Altogen Labs facility is IACUC-regulated and GLP-compliant. Following acclimation to the vivarium environment, mice are sorted according to body mass. The animals are examined daily for tumor appearance and clinical signs. We provide detailed experimental procedures, health reports and data (all-inclusive report is provided to the client that includes methods, results, discussion and raw data along with statistical analysis). Additional services available include collection of tissue, histology, isolation of total protein or RNA and analysis of gene expression.
Following options are available for the MKN-45 xenograft model:
- MKN-45 Tumor Growth Delay (TGD; latency)
- MKN-45 Tumor Growth Inhibition (TGI)
- Dosing frequency and duration of dose administration
- MKN-45 tumor immunohistochemistry
- Gross necropsies and histopathology
- Lipid distribution and metabolic assays
- Imaging studies: Fluorescence-based whole body imaging
