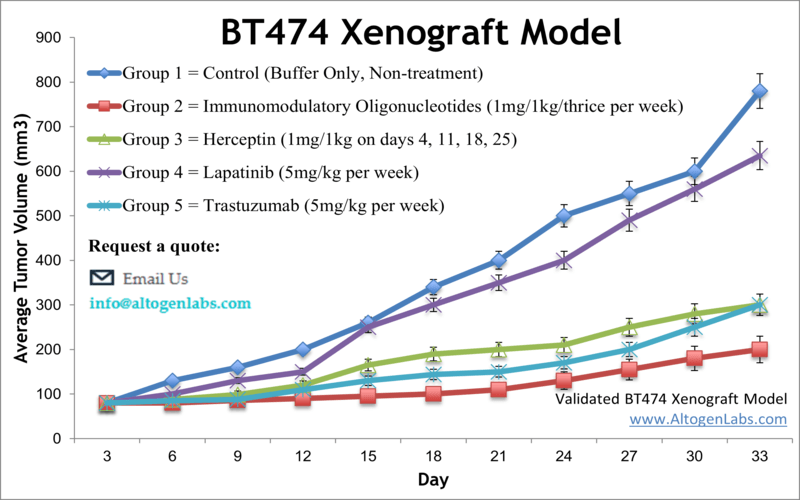
BT474 xenograft model
Invasive ductal carcinoma represents the most common form of breast cancer and is found in about 80 percent of all breast cancer cases. Despite significant improvements in breast cancer management and diagnosis, early detection of breast cancer plays an essential role in the overall treatment. Preclinical studies of the BT474 mouse xenograft model can be used to evaluate anti-cancer therapies. Well-studied breast tumor cell lines are indispensable tools for investigating cell biology of carcinomas. Breast cancer cell lines are extensively employed in preclinical research as molecular models of different types of breast tumors. The BT474 tumorigenic epithelial cell line is isolated by E. Lasfargues and W.G. Coutinho from a solid, invasive ductal carcinoma of the human breast of a 60-year-old Caucasian female patient. The BT474 cell line is aneuploid with a modal chromosome number of 55, as indicated in a 1978 article in Journal of the National Cancer Institute. The BT474 cell line is used to create the CDX (Cell Line Derived Xenograft) BT474 xenograft mouse model. The BT474 breast cancer model can be used to screen anti-cancer therapeutics related to the standard of care drugs (herceptin, lapatinib, trastuzumab) or hormone-induced disease progression (progesterone, norgestrel, norethindrone and MPA).
Examples include the 2015 OncoImmunology study (Zhang et al.) that used the BT474 model to investigate acquired resistance against trastuzumab and develop novel human epidermal growth factor 2 (HER2) antibodies. Their data suggested that blocking the HER2 protein could trigger calreticulin exposure and subsequently cause tumor specific T cell immunity which decreases tumor recurrence. A 1995 study (Slooten et al.) established BT474 as a powerful model for hormone-dependent breast cancer and immune therapy as these cells are oestrogen receptor positive and overexpress c-ErbB-2 protein (more commonly known now as HER2) with the in vivo phenotype of poorly differentiated adenocarcinoma that have high proliferative and turnover rate.
Download Altogen Labs BT474 Xenograft Model PowerPoint Presentation: ![]()
Basic study design
- BT474 cells for injection are maintained under aseptic conditions of exponential growth prior to injection.
- Cells are trypsinized and cell count viability is determined using a trypan blue exclusion assay (98% of cell viability is required). BT474 cell suspension is adjusted to appropriate density.
- Each mouse (NCr-nu/nu mice, 12 w.o.) receive a single subcutaneous injection into the right flank area containing one million cells in a volume of 100 microliters of a Matrigel-BT474 cell suspension.
- The injection sites are palpated up to three times weekly until tumors are established. Tumors are then measured via digital calipers until an average size of 50-150 mm3is established.
- Animals are then randomized into appropriate treatment cohorts and administration of test compound is performed according to the established treatment schedule.
- Mouse weights are recorded 3 times weekly and tumors are measured daily.
- End of study is reached when tumor size reaches 2,000 mm3 or the predetermined size limit per approved IACUC protocol.
- Final necropsy and tissue collections are performed as defined for termination of experiment.
- Tumors are excised, weighed and documented by digital imaging
- Gross necropsies are performed and tissues are collected for downstream analysis.
- Tumors and tissues can be stabilized in RNAlater, snap frozen in LN2or prepared for histology.
Get Instant Quote for
BT474 Xenograft Model
Xenograft animal models are used to assess the effectiveness of experimental test compound against specific types of cancer. New medicines are tested on staged tumor growths that have been engrafted via subcutaneous or orthotopic inoculation in an immunocompromised mouse or rat model. All clinically approved anti-cancer agents have been evaluated with conventional preclinical in vivo models. Xenograft studies can be highly complex, starting with the selection of the appropriate animal model, choice of tumorigenic cell line, administration method, dosing, analysis of tumor growth rates and tumor analysis (histology, mRNA and protein expression levels).
Altogen Labs provides an array of laboratory services using over 30 standard Cell Line Derived Xenograft (CDX) models and over 20 PDX models. Researchers investigating the role of specific proteins or gene products in regulating tumor growth can benefit from development of protein overexpression (genetically engineered to ectopically express proteins, tumor suppressors, or oncogenes) and RNAi cell lines with long term gene silencing. Altogen Labs provides quantitative gene expression analysis of mRNA expression (RT-PCR) and protein expression analysis using the WES system (ProteinSimple).
The dosing of the experimental compound of interest is initiated, for a staged study, when the mean tumor size reaches a specified volume (typically 50-100 mm3). In an unstaged study, the dosing of the compound of interest is initiated immediately after xenografting. Mice are dosed once or twice a day for 28 days (or other desired study duration) via the chosen route of administration. Tumor volume (mm3) is calculated via the “(W x W x L) / 2” formula, where W is tumor width and L is tumor length.
Animal handling and maintenance at the Altogen Labs facility is IACUC-regulated and GLP-compliant. Following acclimatization to the vivarium environment, mice are sorted according to body mass. The animals are examined daily for tumor appearance and clinical signs. We provide detailed experimental procedures, health reports and data (all-inclusive report is provided to the client that includes methods, results, discussion and raw data along with statistical analysis). Additional services available include collection of tissue, histology, isolation of total protein or RNA and analysis of gene expression. Our animal facilities have the flexibility to use specialized food or water systems for inducible gene expression systems.
Following options are available for the BT474 xenograft model:
- BT474 Tumor Growth Delay (TGD; latency)
- BT474 Tumor Growth Inhibition (TGI)
- Dosing frequency and duration of dose administration
- Dosing route (intravenous, intratracheal, continuous infusion, intraperitoneal, intratumoral, oral gavage, topical, intramuscular, subcutaneous, intranasal, using cutting-edge micro-injection techniques and pump-controlled IV injection)
- BT474 tumor immunohistochemistry
- Alternative cell engraftment sites (orthotopic transplantation, tail vein injection and left ventricular injection for metastasis studies, injection into the mammary fat pad, intraperitoneal injection)
- Blood chemistry analysis
- Toxicity and survival (optional: performing a broad health observation program)
- Gross necropsies and histopathology
- Positive control group employing cyclophosphamide, at a dosage of 50 mg/kg administered by intramuscular injection to the control group daily for the study duration
- Lipid distribution and metabolic assays
- Imaging studies: Fluorescence-based whole body imaging, MRI
