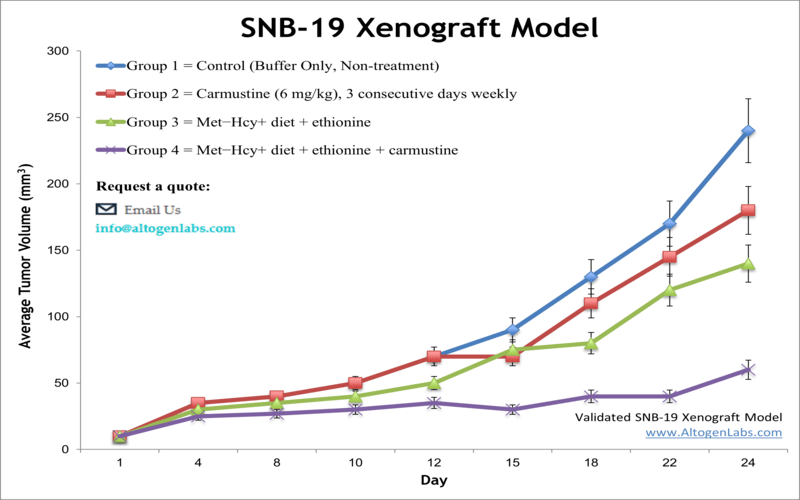
SNB-19 xenograft model
UPDATE: According to ATCC the analysis of short tandem repeats (STRs) indicated that the SNB-19 human glioblastoma cell line exhibits an STR pattern that is indistinguishable from that of the U-373 MG cell line (shown to be a U251MG derivative). Furthermore, SNB-19 and U-373 MG also demonstrate a commonality in their derivative chromosomes. These findings were validated using the original cell stocks held by the American Type Culture Collection (ATCC). According to Cellosaurus SNB-19 cell line has been shown to be a U-251MG derivative.
Prior to recent characterization of SNB-19 cell line (see UPDATE above), this cell line was reported as derived from a glioblastoma multiforme, a highly malignant brain tumor. The American Brain Tumor Association has reported the complete lack of response for select glioblastoma types to treatment. The SNB-19 cell line was utilized in biological research related to human glioblastoma. In a 1988 study, published in Cancer Research Journal, the SNB-19 cell line was the most clonogenic in soft agar and showed the most tumorigenicity in nude mice. Bichat et al. (2000) reported in Clinical Cancer Research the enhanced efficacy of cytotoxic agents with simultaneous methionine depletion especially for treatment of drug-resistant tumors. In this study SNB-19 cells were used as a glioma xenograft model where survival duration was successfully extended with this regimen. A 2015 study (Latocha et al.) used in vitro SNB-19 cells to study the mechanism of action of phenothiazine amine derivatives; they looked at cytotoxic activity, apoptosis induction, effect on proliferation, cell cycle gene expression and total cell oxidative status. SNB-19 cells were used to create the SNB19 xenograft mouse model.
Download Altogen Labs SNB19 Xenograft Model PowerPoint Presentation: ![]()
Basic study design
1. SNB-19 cells used for injection are maintained under conditions of exponential growth prior to injection
2. SNB-19 cells are prepared for injection by trypsinization, and viable cell counts are determined using trypan blue exclusion (98% cell viability required). Cell suspension adjusted to appropriate density
3. Each mouse (athymic BALB/C or NOD/SCID, 10-12 w.o.) receive a subcutaneous injection in the flank of the hind leg of one million cells in a volume of 100 microliters of matrigel-SNB-19 cell suspension
4. The injection sites are palpated three times weekly until tumors are established. Tumors are then measured using digital calipers until they reach an average size of 100-150 mm³.
5. Animals are randomized into treatment cohorts and administration of compound of interest is performed according to the treatment schedule
6. Tumors are measured daily and mouse weights recorded 3 times weekly
7. Animals are euthanized when tumor size reaches 2,000 cubic millimeters or predetermined size limit
8. Necropsy and tissue collection are performed as defined for termination of experiment
9. Tumors are excised, weighed and documented by digital imaging
10. Standard gross necropsies are performed and tissues collected for downstream analysis
11. Tumors and tissues can be snap frozen in LN2 and prepared for histology or gene expression analysis
