SK-N-AS xenograft model
Neuroblastoma is the most common extracranial solid tumor in children; roughly half of the patients experience bone metastasis accompanied by bone pain, leading to poor prognosis where long term survival of high-risk patients is below 40%. The SK-N-AS is hyperdiploid cell line derived from a bone marrow metastasis of a 6-year old female patient. According to a 1989 article in the Journal of Clinical Investigation, SK-N-AS cells taken from human neuroblastoma replicate autonomously due to stimulation by IGF-2. The SK-N-AS cell line produces large amounts of IGF-2 and expresses type 1 insulin-like growth factor receptors. SK-N-AS cells can be useful in the preclinical investigative research for several types of brain cancer. Polo-like kinase 1 (PLK1) is overexpressed in many cancers including high-risk neuroblastoma and is linked to unfavorable patient outcome; a 2011 study by Ackermann et al. demonstrated the strong antitumor activity of the PLK1 inhibitor BI 2536 using this SK-N-AS xenograft mouse model. A 2014 study in the Journal of Bone Oncology by Tsutsuminmoto et al. used the SK-N-AS xenograft model to investigate the mechanism of bone metastasis in neuroblastoma. They found that SK-N-AS cells, but not other neuroblastoma cells, induced expression of the receptor activator of NF-κB ligand, expressed COX-2 mRNA and produced high levels of prostaglandin E2 (PGE2); these phenotypes were suggested to be critical for osteoclastogenesis and developing bone metastases. In 2016, Beadry et al. released a study in the Pedatric Blood Cancer journal using the SK-N-AS xenograft mouse model; this study demonstrated the successful differentiation of high-risk from low-risk patients in addition to active disease from complete response (CR) to very good partial response (VGPR) patients by using serum metabolomics analysis (differences were observed in amino acid, nitrogen, ketosis and carbohydrate metabolism). They concluded that this technique can potentially predict neuroblastoma behavior in humans and allow for improvement and fine-tuning of therapy regimens.
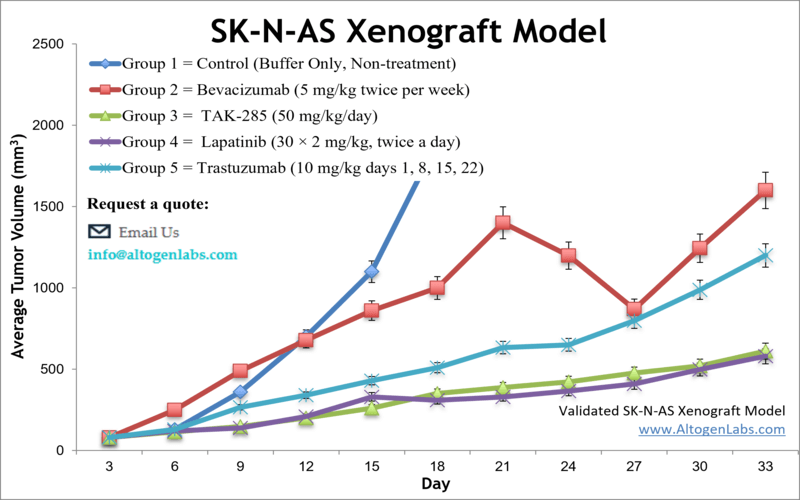
Altogen Labs provides customizable xenotransplantation services for SK-N-AS cells that include comprehensive metabolic assay reports. The SK-N-AS cell line is used to create the CDX (Cell Line Derived Xenograft) SK-N-AS xenograft mouse model. The highly expressing PLK-1 SK-N-AS xenograft is a high-risk neuroblastoma model used to study the anticancer activity of PLK-1 inhibitors (e.g. BI 2536) including mainstream tumor growth inhibitors (e.g. lapatinib, trastuzumab, rosiglitazone).
Download Altogen Labs SKNAS Xenograft Model PowerPoint Presentation: ![]()
Basic study design
- SK-N-AS cells are trypsinized while cell growth is at exponential phase growth. Flow cytometry (Guava PCA) cell viability assay is performed to confirm at least 97-98% cell viability and measure cell concentration. Total suspension concentration is corrected to the cell density needed for injection.
- Athymic nude mice strain (Foxn1nu/Foxn1+) about 10-12 weeks old receive a single 100 µL injection (subcutaneous) in a hind leg containing one million cells (50% Matrigel + SKNAS cell suspension).
- Palpations are performed up to three times a week until tumor establishment. Calipering of tumors are performed to monitor progression of tumor growth. In-life study begins when a size of 50-150 mm3 is reached.
- Animals are grouped into client designed cohorts and the test compounds are administered. Tumors are continuously monitored (daily measurements) along with mouse body weights logged.
- The in-life study is terminated when tumor size reaches the study tumor size limit. As directed by the client, tissues are collected and prepared for histology, frozen or nucleic acids isolated. All resected tumors are digitally imaged and weighed.
Get Instant Quote for
SK-N-AS Xenograft Model
The different types of brain tumor cell lines are astrocytes, neurons, Schwann cell lines, brain endothelial cells, glial, pituitary tumor, retinal, medulloblastoma, and GBM stem cells. Astrocytes are the most abundant cell type in mammalian brains. It shows excellent proliferative ability. It includes glial fibrillary protein. Neuron cells have mature neurons which do not undergo cell division. One way to overcome this is to establish secondary cell lines that are derived from neuronal tumors and have become immortalized. They have the advantage of being grown fairy in cell cultures. Schwann cell lines play an essential role in development, maintenance, function, & regeneration of peripheral nerves. Brain endothelial nerves have the capacity to regulate the passage of the molecules and cells to and from the neural parenchyma and constitute the blood brain barrier. Glial cells, in response to injury, retain the ability to divide. Pituitary tumor cells play a critical role in the development and maintenance of adjacent photoreceptors in the vertebrae retina. Medulloblastoma cells form in the cerebrum, it forms in fetal cells that remain after birth. GBM stem cells differentiate into cells of all three neutral lineages including cells expressing markers of immature neurons. Xenograft animal models are used to assess the effectiveness of experimental test compounds against specific types of cancer. Novel medicines are tested on staged tumor growths that have been engrafted via subcutaneous or orthotopic inoculation in an immunocompromised mouse or rat model (Nude or NOD/SCID). All clinically approved anti-cancer agents have been evaluated with conventional preclinical in vivo models.
SK-N-AS is a human neuroblastoma cell line that is often used in preclinical research as a model for studying the biology of neuroblastoma and testing potential treatments for the disease. SK-N-AS cells were originally derived from a bone marrow metastasis in a patient with neuroblastoma. SK-N-AS xenograft studies performed to study tumor growth and response to treatment in vivo. Xenograft studies can be highly complex, starting with the selection of the appropriate animal model, choice of tumorigenic cell line, administration method, dosing, analysis of tumor growth rates and tumor analysis (histology, mRNA and protein expression levels). Altogen Labs provides an array of laboratory services using over 120 CDX and PDX models. Researchers investigating the role of specific proteins or gene products in regulating tumor growth can benefit from development of protein overexpression (genetically engineered to ectopically express proteins, tumor suppressors, or oncogenes) and RNAi cell lines with long term gene silencing. Animal handling and maintenance at the Altogen Labs facilities are IACUC regulated and GLP-compliant. We provide detailed experimental procedures, health reports and data (all-inclusive report is provided to the client that includes methods, results, discussion and raw data along with statistical analysis).
Following options are available for the SKNAS xenograft model:
- SK-N-AS Tumor Growth Delay (TGD; latency)
- SK-N-AS Tumor Growth Inhibition (TGI)
- Dosing frequency and duration of dose administration
- Dosing route (intravenous, intratracheal, continuous infusion, intraperitoneal, intratumoral, oral gavage, topical, intramuscular, subcutaneous, intranasal, using cutting-edge micro-injection techniques and pump-controlled IV injection)
- SK-N-AS tumor immunohistochemistry
- Alternative cell engraftment sites (orthotopic transplantation)
- Blood chemistry analysis
- Toxicity and survival (optional: performing a broad health observation program)
- Gross necropsies and histopathology
